临床试验英语词汇
临床试验英语词汇

专业术语、缩略语中英对照表缩略语英文全称中文全称ABE Average Bioequivalence 平均生物等效性AC Active control 阳性对照ADE Adverse Drug Event 药物不良事件ADR Adverse Drug Reaction 药物不良反应AE Adverse Event 不良事件AI Assistant Investigator 助理研究者ALB Albumin 白蛋白ALD Approximate Lethal Dose 近似致死剂量ALP Alkaline phosphatase 碱性磷酸酶ALT Alanine aminotransferase 丙氨酸转氨酶ANDA Abbreviated New Drug Application 简化新药申请ANOV A Analysis of variance 方差分析AST Aspartate aminotransferase 天冬氨酸转氨酶ATR Attenuated total reflection 衰减全反射法BA Bioavailability 生物利用度BE Bioequivalence 生物等效性BMI Body Mass Index 体质指数BUN Blood urea nitrogen 血尿素氮CATD Computer-assisted trial design 计算机辅助试验设计CDER Center of Drug Evaluation and Research 药品评价和研究中心CFR Code of Federal Regulation 美国联邦法规CI Co-Investigator 合作研究者CI Confidence Interval 可信区间COI Coordinating Investigator 协调研究者CRC Clinical Research Coordinator 临床研究协调者CRF Case Report/Record Form 病历报告表/病例记录表CRO Contract Research Organization 合同研究组织CSA Clinical Study Application 临床研究申请CTA Clinical Trial Application 临床试验申请CTP Clinical Trial Protocol 临床试验方案CTR Clinical Trial Report 临床试验报告CTX Clinical Trial Exemption 临床试验免责CHMP Committee for Medicinal 人用药委会Products for Human UseDSC Differential scanning 差示扫描热量计DSMB Data Safety and monitoring Board 数据安全及监控委员会EDC Electronic Data Capture 电子数据采集系统EDP Electronic Data Processing 电子数据处理系统EWP Europe Working Party 欧洲工作组FDA Food and Drug Administration 美国食品与药品管理局FR Final Report 总结报告GCP Good Clinical Practice 药物临床试验质量管理规范GCP Good Laboratory Practice 药物非临床试验质量管理规范GLU Glucose 葡萄糖GMP Good Manufacturing Practice 药品生产质量管理规范HEV Health economic evaluation 健康经济学评价IB Investigator’s Brochure研究者手册IBE IndividualBioequivalence 个体生物等效性IC Informed Consent 知情同意ICF Informed Consent Form 知情同意书ICH International Conference on Harmonization 国际协调会议IDM Independent Data Monitoring 独立数据监察IDMC Independent Data Monitoring Committee 独立数据监察委员会IEC Independent Ethics Committee 独立伦理委员会IND Investigational New Drug 新药临床研究IRB Institutional Review Board 机构审查委员会ITT Intention-to –treat 意向性分析IVD In Vitro Diagnostic 体外诊断IVRS Interactive Voice Response System 互动语音应答系统LD50 Medial lethal dose 半数致死剂量LLOQ Lower Limit of quantitation 定量下限LOCF Last observation carry forward 最接近一次观察的结转LOQ Limit of Quantitation 检测限MA Marketing Approval/Authorization 上市许可证MCA Medicines Control Agency 英国药品监督局MHW Ministry of Health and Welfare 日本卫生福利部MRT Mean residence time 平均滞留时间MTD Maximum Tolerated Dose 最大耐受剂量ND Not detectable 无法定量NDA New Drug Application 新药申请NEC New Drug Entity 新化学实体NIH National Institutes of Health 国家卫生研究所(美国)NMR Nuclear Magnetic Resonance 核磁共振PD Pharmacodynamics 药效动力学PI Principal Investigator 主要研究者PK Pharmacokinetics 药物动力学PL Product License 产品许可证PMA Pre-market Approval (Application) 上市前许可(申请)PP Per protocol 符合方案集PSI Statisticians in the Pharmaceutical Industry 制药业统计学家协会QA Quality Assurance 质量保证QAU Quality Assurance Unit 质量保证部门QC Quality Control 质量控制QWP Quality Working Party 质量工作组RA Regulatory Authorities 监督管理部门REV Revision 修订SA Site Assessment 现场评估SAE Serious Adverse Event 严重不良事件SAP Statistical Analysis Plan 统计分析计划SAR Serious Adverse Reaction 严重不良反应SD Source Data/Document 原始数据/文件SD Subject Diary 受试者日记SDV Source Data Verification 原始数据核准SEL Subject Enrollment Log 受试者入选表SFDA State Food and Drug Administration 国家食品药品监督管理局SI Sponsor-Investigator 申办研究者SI Sub-investigator 助理研究者SIC Subject Identification Code 受试者识别代码SOP Standard Operating Procedure 标准操作规程SPL Study Personnel List 研究人员名单SSL Subject Screening Log 受试者筛选表T&R Test and Reference Product 受试和参比试剂T-BIL Total Bilirubin 总胆红素T-CHO Total Cholesterol 总胆固醇TG Thromboglobulin 血小板球蛋白Tmax Time of maximum concentration 达峰时间TP Total proteinum 总蛋白UAE Unexpected Adverse Event 预料外不良事件WHO World Health Organization 世界卫生组织WHO- WHO International Conference WHO 国际药品管理当局会议ICDR A of Drug Regulatory AuthoritiesAberrant result 异常结果Absorption phase 吸收相Absorption 吸收Accuracy 准确度Accurate 精密度Administer 给药Amendment修正案Approval 批准Assess 估计Audit Report 稽查报告Audit 稽查Auditor 稽查员Analytical run/batch:分析批Benefit 获益Bias 偏性,偏倚Bioequivalence 生物等效Biosimilar /Follow-on biologics 生物仿制药Blank Control 空白对照Blind codes 编制盲底Blind review 盲态检查/盲态审核Blinding method 盲法Blinding/masking 盲法/设盲Block size 每段的长度Block 层/分段BCS 生物药剂学分类系统精品文库Carryover effect 延滞效应Case history 病历Clinical equivalence 临床等效性Clinical study 临床研究Clinical Trial Report 临床试验报告Comparison 对照Compensation 补偿,赔偿金Compliance 依从性Concomitant 伴随的Conduct 行为Confidence level 置信水平Consistency test 一致性检验Contract/ agreement 协议/合同Control group 对照组Coordinating Committee 协调委员会Crossover design 交叉设计Cross-over Study 交叉研究Cure 痊愈Data management 数据管理Descriptive statistical analysis 描述性统计分析Dichotomies 二分类Dispense 分布Diviation 偏差Documentation 记录/文件Dosage forms 剂型Dose dumping 剂量倾卸(药物迅速释放入血而达到危险浓度)Dose-reaction relation 剂量-反应关系Double blinding 双盲精品文库Double dummy 双模Drop out 脱落Effectiveness 疗效Elimination phase 消除相Emergency envelope 应急信件Enantiomers 对映体End point 终点Endpoint criteria/ measurement 终点指标Enterohepatic recycling 肠肝循环Essential Documentation 必需文件Ethical 伦理的Ethics committee 伦理委员会Evaluate 评估Exclusion Criteria 排除标准Excretion 排泄Expedite 促进Extrapolated 外推的Essentially similar product:基本相似药物Factorial design 析因设计Failure 无效,失败Finacing 财务,资金Final point 终点First pass metabolism 首过代谢Fixed-dose procedure 固定剂量法Full analysis set 全分析集GC-FTIR 气相色谱-傅利叶红外联用GC-MS 气相色谱-质谱联用Generic drug 通用名药精品文库Gene mutation 基因突变Genotoxicity tests 生殖毒性试验Global assessment variable 全局评价变量Group sequential design 成组序贯设计Hypothesis test 假设检验Highly permeable:高渗透性Highly soluble:高溶解度Highly variable drug:高变异性药物Highly:Variable Drug 高变异性药物HVDP:高变异药物制剂Identification 鉴别,身份证Improvement 好转In vitro 体外In vivo 体内Inclusion Criteria 入选表准Information Gathering 信息收集Initial Meeting 启动会议Inspection 检察/视察Institution Inspection 机构检察Instruction 指令,说明Integrity 完整,正直Intercurrent 中间发生的,间发的Inter-individual variability 个体间变异性Interim analysis 期中分析Investigational Product 试验药物Investigator 研究者Involve 引起,包括IR 红外吸收光谱精品文库Innovator Product:原创药Ka 吸收速率常LC-MS 液相色谱-质谱联用logarithmic transformation 对数转换Logic check 逻辑检查Lost of follow up 失访Mask 面具,掩饰Matched pair 匹配配对Metabolism 代谢Missing value 缺失值Mixed effect model 混合效应模式Modified release products 改良释放剂型Monitor 监查员Monitoring Plan 监察计划Monitoring Report 监察报告MS-MS 质谱-质谱联用Multi-center Trial 多中心试验Negative 阴性,否定的Non-clinical Study 非临床研究Non-inferiority 非劣效性Non-Linear Pharmacokinetics 非线性药代动力学Non-parametric statistics 非参数统计方法NTID:窄治疗指数制剂Obedience 依从性Open-blinding 非盲Open-label 非盲Original Medical Record 原始医疗记录Outcome Assessment 结果评价精品文库Outcome measurement 结果指标Outlier 离群值OIP 经口服吸收药物Parallel group design 平行组设计Parameter estimation 参数估计Parametric statistics 参数统计方法Patient file 病人档案Patient History 病历Per protocol,PP 符合方案集Permeability 渗透性Pharmacodynamic characteristics 药效学特征Pharmacokinetic characteristics 药代学特征Placebo Control 安慰剂对照Placebo 安慰剂Polytomies 多分类Post-dosing postures 给药后坐姿Potential 潜在的Power 检验效能Precision 精密度Preclinical Study 临床前研究Precursor 母体前体Premature 过早的,早发Primary endpoint 主要终点Primary variable 主要变量Prodrug 药物前体Protocol amendment 方案补正Protocol Amendments 修正案Protocol 试验方案精品文库Quality Control Sample:质控样品Rapidly dissolving:快速溶出Racemates 外消旋物Randomization 随机/随机化Range check 范围检Rating scale 量表Recruit 招募,新会员Replication 可重复Retrieval 取回,补修Revise 修正Risk 风险Run in 准备期Safety evaluation 安全性评价Safety set 安全性评价的数据集Sample Size 样本量、样本大小Sampling schedules 采血计划Scale of ordered categorical ratings 有序分类指标Secondary variable 次要变量Sequence 试验次序Seriousness 严重性Severity 严重程度Significant level 检验水准Simple randomization 简单随机Single Blinding 单盲Site audit 试验机构稽查Solubility 溶解度Specificity 特异性Specify 叙述,说明精品文库Sponsor-investigator 申办研究者Standard curve 标准曲线Statistical model 统计模型Statistical tables 统计分析表Steady state 稳态Storage 储存Stratified 分层Study Audit 研究稽查Study Site 研究中心Subgroup 亚组Sub-investigator 助理研究者Subject Enrollment Log 受试者入选表Subject Enrollment 受试者入选Subject Identification Code List 受试者识别代码表Subject Recruitment 受试者招募Subject Screening Log 受试者筛选表Subject 受试者Submit 交付,委托Superiority 检验Supplemental 增补的Supra-bioavailability 超生物利用度(试验药的生物利用度大于对照药)Survival analysis 生存分析System Audit 系统稽查SmPC:药品说明书Standard Sample:标准样品Target variable 目标变量Treatment group 试验组Trial error 试验误差精品文库Trial Initial Meeting 试验启动会议Trial Master File 试验总档案Trial Objective 试验目的Trial site 试验场所Triple Blinding 三盲Two one-side test 双单侧检验Therapeutic equivalence:治疗等效性Un-blinding 破盲/揭盲Verify 查证、核实Visual analogy scale 直观类比打分法Vulnerable subject 弱势受试者Wash-out Period 洗脱期Well-being 福利,健康Withdraw 撤回,取消药代动力学参数Ae(0-t):给药到t时尿中排泄的累计原形药。
临床试验英语词汇

临床试验英语词汇————————————————————————————————作者:————————————————————————————————日期:ﻩ专业术语、缩略语中英对照表缩略语英文全称中文全称ABE Average Bioequivalence平均生物等效性ACActivecontrol 阳性对照ADEAdverseDrug Event药物不良事件ADR AdverseDrug Reaction 药物不良反应AE Adverse Event 不良事件AIAssistant Investigator 助理研究者ALB Albumin白蛋白ALDApproximateLethal Dose近似致死剂量ALP Alkaline phosphatase 碱性磷酸酶ALT Alanineaminotransferase 丙氨酸转氨酶ANDAAbbreviated New Drug Application 简化新药申请ANOV A Analysis of variance方差分析AST Aspartate aminotransferase天冬氨酸转氨酶ATR Attenuatedtotal reflection 衰减全反射法BABioavailability生物利用度BE Bioequivalence生物等效性BMIBodyMass Index 体质指数BUN Blood ureanitrogen血尿素氮CATDComputer-assistedtrial design计算机辅助试验设计CDER Center of DrugEvaluation andResearch药品评价和研究中心CFRCode ofFederal Regulation美国联邦法规CI Co-Investigator合作研究者CI Confidence Interval可信区间COI Coordinating Investigator 协调研究者CRC Clinical ResearchCoordinator临床研究协调者CRF Case Report/Record Form病历报告表/病例记录表CRO Contract ResearchOrganization 合同研究组织CSA Clinical Study Application临床研究申请CTA Clinical Trial Application 临床试验申请CTPClinical Trial Protocol临床试验方案CTR ClinicalTrial Report 临床试验报告CTXClinical Trial Exemption临床试验免责CHMP Committee for Medicinal 人用药委会Products for Human UseDSCDifferentialscanning差示扫描热量计DSMBData Safety andmonitoring Board数据安全及监控委员会EDCElectronic Data Capture电子数据采集系统EDP Electronic Data Processing 电子数据处理系统EWPEurope Working Party欧洲工作组FDA Foodand Drug Administration 美国食品与药品管理局FR FinalReport 总结报告GCP GoodClinicalPractice药物临床试验质量管理规范GCP Good Laboratory Practice 药物非临床试验质量管理规范GLU Glucose葡萄糖GMP GoodManufacturing Practice 药品生产质量管理规范HEV Healtheconomic evaluation健康经济学评价IB Investigator’s Brochure研究者手册IBE IndividualBioequivalence个体生物等效性IC Informed Consent 知情同意ICF InformedConsentForm知情同意书ICH International Conference onHarmonization 国际协调会议IDMIndependentData Monitoring 独立数据监察IDMC IndependentData Monitoring Committee 独立数据监察委员会IEC Independent EthicsCommittee 独立伦理委员会IND Investigational New Drug新药临床研究IRB Institutional Review Board机构审查委员会ITT Intention-to –treat 意向性分析IVD In VitroDiagnostic体外诊断IVRS InteractiveVoice Response System 互动语音应答系统LD50 Mediallethaldose半数致死剂量LLOQLower Limit ofquantitation 定量下限LOCF Last observation carry forward 最接近一次观察的结转LOQ Limitof Quantitation检测限MA Marketing Approval/Authorization 上市许可证MCA Medicines Control Agency 英国药品监督局MHW Ministryof HealthandWelfare 日本卫生福利部MRT Mean residencetime 平均滞留时间MTD MaximumToleratedDose 最大耐受剂量ND Not detectable无法定量NDANew Drug Application 新药申请NEC New DrugEntity 新化学实体NIH NationalInstitutesof Health国家卫生研究所(美国)NMR NuclearMagneticResonance核磁共振PD Pharmacodynamics 药效动力学PI Principal Investigator 主要研究者PK Pharmacokinetics药物动力学PLProduct License 产品许可证PMA Pre-market Approval (Application)上市前许可(申请)PP Perprotocol符合方案集PSI Statisticians inthe PharmaceuticalIndustry制药业统计学家协会QA QualityAssurance 质量保证QAU QualityAssurance Unit 质量保证部门QC Quality Control 质量控制QWP QualityWorking Party 质量工作组RARegulatory Authorities 监督管理部门REV Revision修订SA Site Assessment 现场评估SAESeriousAdverse Event 严重不良事件SAPStatistical Analysis Plan 统计分析计划SAR Serious Adverse Reaction严重不良反应SD Source Data/Document 原始数据/文件SD Subject Diary受试者日记SDV Source Data Verification 原始数据核准SELSubject Enrollment Log 受试者入选表SFDA State FoodandDrug Administration 国家食品药品监督管理局SI Sponsor-Investigator 申办研究者SI Sub-investigator 助理研究者SICSubjectIdentification Code受试者识别代码SOP Standard Operating Procedure标准操作规程SPLStudy PersonnelList 研究人员名单SSLSubjectScreening Log 受试者筛选表T&RTestand Reference Product 受试和参比试剂T-BIL Total Bilirubin 总胆红素T-CHO Total Cholesterol总胆固醇TG Thromboglobulin 血小板球蛋白Tmax Time of maximum concentration 达峰时间TP Total proteinum 总蛋白UAEUnexpected Adverse Event 预料外不良事件WHO World Health Organization世界卫生组织WHO-WHOInternational Conference WHO 国际药品管理当局会议ICDR A of Drug RegulatoryAuthoritiesAberrant result 异常结果Absorption phase 吸收相Absorption 吸收Accuracy 准确度Accurate 精密度Administer 给药Amendment修正案Approval批准Assess 估计AuditReport 稽查报告Audit 稽查Auditor 稽查员Analytical run/batch:分析批Benefit 获益Bias 偏性,偏倚Bioequivalence 生物等效Biosimilar/Follow-on biologics 生物仿制药Blank Control 空白对照Blind codes编制盲底Blind review 盲态检查/盲态审核Blindingmethod 盲法Blinding/masking盲法/设盲Blocksize 每段的长度Block 层/分段BCS生物药剂学分类系统Carryover effect延滞效应Case history病历Clinical equivalence临床等效性Clinical study 临床研究Clinical Trial Report 临床试验报告Comparison对照Compensation补偿,赔偿金Compliance 依从性Concomitant 伴随的Conduct行为Confidence level置信水平Consistency test一致性检验Contract/ agreement 协议/合同Control group对照组CoordinatingCommittee 协调委员会Crossover design 交叉设计Cross-over Study 交叉研究Cure 痊愈Datamanagement数据管理Descriptivestatisticalanalysis描述性统计分析Dichotomies二分类Dispense 分布Diviation偏差Documentation 记录/文件Dosage forms剂型Dose dumping 剂量倾卸(药物迅速释放入血而达到危险浓度) Dose-reaction relation 剂量-反应关系Doubleblinding 双盲Double dummy双模Dropout 脱落Effectiveness疗效Eliminationphase 消除相Emergency envelope 应急信件Enantiomers 对映体End point 终点Endpointcriteria/measurement终点指标Enterohepatic recycling 肠肝循环Essential Documentation必需文件Ethical伦理的Ethicscommittee 伦理委员会Evaluate 评估Exclusion Criteria排除标准Excretion排泄Expedite促进Extrapolated外推的Essentially similarproduct:基本相似药物Factorialdesign析因设计Failure 无效,失败Finacing 财务,资金Final point终点First passmetabolism 首过代谢Fixed-dose procedure 固定剂量法Fullanalysis set 全分析集GC-FTIR气相色谱-傅利叶红外联用GC-MS气相色谱-质谱联用Genericdrug通用名药Genemutation基因突变Genotoxicity tests 生殖毒性试验Global assessmentvariable全局评价变量Group sequential design 成组序贯设计Hypothesis test假设检验Highly permeable:高渗透性Highlysoluble:高溶解度Highly variable drug:高变异性药物Highly:VariableDrug 高变异性药物HVDP:高变异药物制剂Identification 鉴别,身份证Improvement好转Invitro 体外In vivo 体内Inclusion Criteria 入选表准InformationGathering信息收集InitialMeeting启动会议Inspection 检察/视察Institution Inspection 机构检察Instruction 指令,说明Integrity 完整,正直Intercurrent 中间发生的,间发的Inter-individual variability 个体间变异性Interim analysis 期中分析Investigational Product试验药物Investigator 研究者Involve引起,包括IR红外吸收光谱Innovator Product:原创药Ka吸收速率常LC-MS液相色谱-质谱联用logarithmic transformation 对数转换Logiccheck 逻辑检查Lostof followup失访Mask面具,掩饰Matched pair匹配配对Metabolism 代谢Missingvalue 缺失值Mixedeffect model 混合效应模式Modified releaseproducts改良释放剂型Monitor 监查员Monitoring Plan 监察计划Monitoring Report 监察报告MS-MS质谱-质谱联用Multi-center Trial多中心试验Negative 阴性,否定的Non-clinical Study 非临床研究Non-inferiority 非劣效性Non-Linear Pharmacokinetics非线性药代动力学Non-parametricstatistics非参数统计方法NTID:窄治疗指数制剂Obedience 依从性Open-blinding非盲Open-label 非盲Original Medical Record 原始医疗记录Outcome Assessment 结果评价Outcome measurement结果指标Outlier 离群值OIP 经口服吸收药物Parallel group design 平行组设计Parameter estimation参数估计Parametricstatistics参数统计方法Patient file 病人档案Patient History 病历Per protocol,PP符合方案集Permeability渗透性Pharmacodynamiccharacteristics药效学特征Pharmacokinetic characteristics药代学特征Placebo Control 安慰剂对照Placebo安慰剂Polytomies多分类Post-dosing postures给药后坐姿Potential 潜在的Power 检验效能Precision 精密度Preclinical Study临床前研究Precursor母体前体Premature 过早的,早发Primary endpoint 主要终点Primary variable主要变量Prodrug 药物前体Protocolamendment方案补正Protocol Amendments修正案Protocol试验方案Quality Control Sample:质控样品Rapidly dissolving:快速溶出Racemates 外消旋物Randomization 随机/随机化Range check 范围检Rating scale量表Recruit招募,新会员Replication可重复Retrieval取回,补修Revise 修正Risk 风险Run in 准备期Safety evaluation安全性评价Safety set安全性评价的数据集SampleSize样本量、样本大小Samplingschedules采血计划Scaleoforderedcategorical ratings有序分类指标Secondaryvariable 次要变量Sequence 试验次序Seriousness严重性Severity严重程度Significant level 检验水准Simple randomization简单随机Single Blinding 单盲Siteaudit试验机构稽查Solubility溶解度Specificity 特异性Specify叙述,说明Sponsor-investigator申办研究者Standard curve 标准曲线Statistical model 统计模型Statistical tables 统计分析表Steadystate 稳态Storage储存Stratified 分层StudyAudit研究稽查Study Site研究中心Subgroup 亚组Sub-investigator 助理研究者Subject EnrollmentLog受试者入选表SubjectEnrollment 受试者入选Subject IdentificationCode List受试者识别代码表Subject Recruitment 受试者招募Subject Screening Log 受试者筛选表Subject 受试者Submit交付,委托Superiority 检验Supplemental增补的Supra-bioavailability 超生物利用度(试验药的生物利用度大于对照药)Survival analysis生存分析System Audit 系统稽查SmPC:药品说明书Standard Sample:标准样品Target variable目标变量Treatment group试验组Trial error 试验误差TrialInitial Meeting试验启动会议Trial Master File 试验总档案Trial Objective试验目的Trial site 试验场所TripleBlinding 三盲Twoone-side test 双单侧检验Therapeuticequivalence:治疗等效性Un-blinding 破盲/揭盲Verify查证、核实Visual analogy scale 直观类比打分法Vulnerablesubject弱势受试者Wash-out Period 洗脱期Well-being 福利,健康Withdraw撤回,取消药代动力学参数Ae(0-t):给药到t时尿中排泄的累计原形药。
临床试验(Clinical Trial)或临床研究(Clinical Study)常用词汇

临床试验(Clinical Trial)或临床研究(Clinical Study)常用词汇全名为International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use,人用药品注册技术规定国际协调会议,是欧洲、日本及美国三方药品管理当局及三方制药企业管理机构,与1990年共同发起,对三方国家人用药品注册技术规定的现存差异进行协调的国际协调组织。
其主要目的是希望在不影响药物品质、安全性及有效性的原则下,能够加速全球新药的研发及上市。
一来,减少新药研发的高昂成本;二来加速新药上市,让病人及早获得较新、较好的治疗。
自1991年11月于比利时布鲁塞尔召开第一届会议后,历经1993年10月美国奥兰多的ICH2,1995年11月日本横滨的ICH3,以及1997年7月比利时布鲁塞尔的ICH4总计有36个议题已完成统一,9个议题完成初稿,所有这些议题涵盖了新药研发乃至上市过程中最为重要的“安全性、质量、有效性和多学科性”四大领域。
GCP Good Clinical Practice,药品临床试验管理规范(GCP)是临床试验全过程的标准规定,制定GCP的目的在于保证临床试验过程的规范,结果科学可靠,保护受试者的权益并保障其安全。
CRO Contract Research Organization(CRO),合同研究组织,是通过合同形式向制药企业提供新药临床研究服务的专业公司。
CRO可在短时间内组织起一个具有高度专业化的和具有丰富临床经验的临床研究队伍,并能降低整个制药企业的管理费用。
SMO Site Management Organization, 现场管理工作的查核机构,不涉及试验和统计的人员组成,体征或实验检查数据的文件。
CRF Case Report Form, 病例报告表,是在临床试验中用以记录每一名受试者在试验过程中的症状、体征或实验室检查数据的文件。
临床试验相关英文及缩写
35 ALP-Alkaline Phosphatase 碱性磷酸酶 36 TB- Total Bilirubin 总胆红素 37 LDH 乳酸脱氢酶 38 GGT 谷氨酰转氨酶 39 Glucose 葡萄糖 40 Blood Coagulation 凝血 41 PT-Prothrombin Time 凝血酶原时间 42 PT-INR 凝血酶原时间-国际标准化比值 43 APTT 活化部分凝血活酶时间 药物相关 IP-Investigational Product 研究产品 CM -Concomitant Medication 合并用药 Drug Receive 药物接收 Drug Storage 药物储存 Drug Dispense 药物分发 Drug Return 药物返还 Drug Destroy 药物销毁 Quantity 数量 Medication No. 研究药品编号 Packaging 包装 Labeling 标签 Drug Accountability 药物清点 Drug Administration 药物使用 Overdose 药物过量 Dose Reduce 剂量降低 Dose Interrupt 剂量中断(暂停) Dose Discontinuation 剂量终止(完全终止) 不良反应相关 ADR- Adverse Drug Reaction 药物不良反应 AE- Adverse Event 不良事件 SAE Serious Adverse Event 严重不良事件 Seriousness 严重性(性质) Severity 严重程度(程度) Grade 级别 Results in death 导致死亡 Life threatening 危急生命 Requires or prolongs hospitalization 导致或延长住院时间 Congenital anomaly/Birth defect 先天畸形/出生缺陷
临床试验术语英汉对照
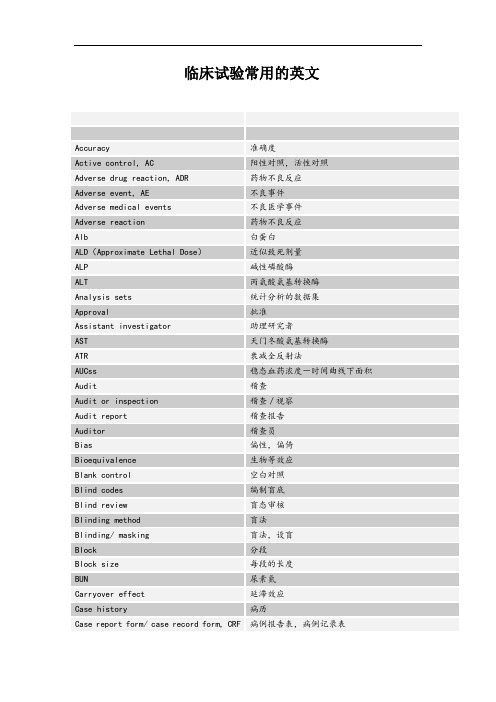
临床试验常用的英文Accuracy准确度Active control, AC阳性对照,活性对照Adverse drug reaction, ADR药物不良反应Adverse event, AE不良事件Adverse medical events不良医学事件Adverse reaction药物不良反应Alb白蛋白ALD(Approximate Lethal Dose)近似致死剂量ALP碱性磷酸酶ALT丙氨酸氨基转换酶Analysis sets统计分析的数据集Approval批准Assistant investigator助理研究者AST天门冬酸氨基转换酶ATR衰减全反射法AUCss稳态血药浓度-时间曲线下面积Audit稽查Audit or inspection稽查/视察Audit report稽查报告Auditor稽查员Bias偏性,偏倚Bioequivalence生物等效应Blank control空白对照Blind codes编制盲底Blind review盲态审核Blinding method盲法Blinding/ masking盲法,设盲Block分段Block size每段的长度BUN尿素氮Carryover effect延滞效应Case history病历Case report form/ case record form, CRF病例报告表,病例记录表Categorical variable分类变量Cav平均浓度CL清除率Clinical equivalence临床等效应Clinical study临床研究Clinical study report临床试验的总结报告Clinical trial临床试验Clinical trial application, CTA临床试验申请Clinical trial exemption, CTX临床试验免责Clinical trial protocol, CTP临床试验方案Clinical trial/ study report临床试验报告C max峰浓度Co-investigator合作研究者Comparison对照Compliance依从性Composite variable复合变量Computer-assisted trial design, CATD计算机辅助试验设计Confidence interval可信区间Confidence level置信水平Consistency test一致性检验Contract research organization, CRO合同研究组织Contract/ agreement协议/合同Control group对照组Coordinating committee协调委员会Crea肌酐CRF(case report form)病例报告表Crossover design交叉设计Cross-over study交叉研究Css稳浓度Cure痊愈Data management数据管理Database建立数据库Descriptive statistical analysis描述性统计分析DF波动系统Dichotomies二分类Diviation偏差Documentation记录/文件Dose-reaction relation剂量-反应关系Double blinding双盲Double dummy双模拟Double dummy technique双盲双模拟技术Double-blinding双盲Drop out脱落Effectiveness疗效Electronic data capture, EDC电子数据采集系统Electronic data processing, EDP电子数据处理系统Emergency envelope应急信件End point终点Endpoint criteria/ measurement终点指标Equivalence等效性Essential documentation必须文件Ethics committee伦理委员会Excellent显效Exclusion criteria排除标准Factorial design析因设计Failure无效,失败Final point终点Fixed-dose procedure固定剂量法Forced titration强制滴定Full analysis set全分析集GC-FTIR气相色谱-傅利叶红外联用GC-MS气相色谱-质谱联用Generic drug通用名药Global assessment variable全局评价变量GLU血糖Good clinical practice, GCP药物临床试验质量管理规范Good manufacture practice, GMP药品生产质量管理规范药物非临床研究质量管理规范Good non-clinical laboratory practice,GLPGroup sequential design成组序贯设计Hypothesis test假设检验International Conference of Harmonization, ICH人用药品注册技术要求国际技术协调会,国际协调会议Improvement好转Inclusion criteria入选标准Independent ethics committee, IEC独立伦理委员会Information consent form, ICF知情同意书Information gathering信息收集Informed consent, IC知情同意Initial meeting启动会议Inspection视察/检查Institution inspection机构检查Institution review board, IBR机构审查委员会Intention to treat意向治疗(-- 临床领域)Intention-to –treat, ITT意向性分析(-统计学)Interim analysis期中分析Investigator研究者Investigator’s brochure, IB研究者手册IR红外吸收光谱Ka吸收速率常Last observation carry forward, LOCF最接近一次观察的结转LC-MS液相色谱-质谱联用LD50半数致死剂量Logic check逻辑检查LOQ (Limit of Quantitation)定量限LOCF, Last observation carry forward最近一次观察的结转Lost of follow up失访Marketing approval/ authorization上市许可证Matched pair匹配配对Missing value缺失值Mixed effect model混合效应模式Monitor监查员Monitoring监查Monitoring report监查报告MRT平均滞留时间MTD(Maximum Tolerated Dose)最大耐受剂量Multi-center trial多中心试验New chemical entity, NCE新化学实体New drug application, NDA新药申请NMR核磁共振谱Non-clinical study非临床研究Non-inferiority非劣效性Non-parametric statistics非参数统计方法Obedience依从性ODR旋光光谱Open-blinding非盲Open-label非盲Optional titration随意滴定Original medical record原始医疗记录Outcome结果Outcome assessment结果指标评价Outcome measurement结果指标Outlier离群值Parallel group design平行组设计Parameter estimation参数估计Parametric statistics参数统计方法Patient file病人档案Patient history病历Per protocol, PP符合方案集Placebo安慰剂Placebo control安慰剂对照Polytomies多分类Power检验效能Precision精密度Preclinical study临床前研究Primary endpoint主要终点Primary variable主要变量Principal investigator主要研究者Principle investigator, PI主要研究者Product license, PL产品许可证Protocol试验方案Protocol amendment方案补正Quality assurance unit, QAU质量保证部门Quality assurance, QA质量保证Quality control, QC质量控制Query list, query form应用疑问表Randomization随机化Range check范围检查Rating scale量表Regulatory authorities, RA监督管理部门Replication可重复RSD日内和日间相对标准差Run in准备期Safety evaluation安全性评价Safety set安全性评价的数据集Sample size样本量,样本大小Scale of ordered categorical ratings有序分类指标Secondary variable次要变量Sequence试验次序Serious adverse event, SAE严重不良事件Serious adverse reaction, SAR严重不良反应Seriousness严重性Severity严重程度Significant level检验水准Simple randomization简单随机Single-blinding单盲Site audit试验机构稽查Source data verification, SDV原始数据核准Source data, SD原始数据Source document, SD原始文件Specificity特异性Sponsor申办者Sponsor-investigator申办研究者Standard curve标准曲线Standard operating procedure, SOP标准操作规程Statistic统计量Statistical analysis plan, SAP统计分析计划Statistical model统计模型Statistical tables统计分析表Stratified分层Study audit研究稽查Subgroup亚组Sub-investigator助理研究者Subject受试者Subject diary受试者日记Subject enrollment受试者入选Subject enrollment log受试者入选表Subject identification code, SIC受试者识别代码Subject recruitment受试者招募Subject screening log受试者筛选表Superiority检验Survival analysis生存分析SXRD单晶X-射线衍射System audit系统稽查T1/2消除半衰期Target variable目标变量T-BIL总胆红素T-CHO总胆固醇TG热重分析TLC、HPLC制备色谱T max峰时间TP总蛋白Transformation变量变换Treatment group试验组Trial error试验误差Trial master file试验总档案Trial objective试验目的Trial site试验场所Triple blinding三盲Two one-side test双单侧检验Unblinding揭盲Unexpected adverse event, UAE预料外不良事件UV-VIS紫外-可见吸收光谱Variability变异Variable变量Visual analogy scale直观类比打分法Visual check人工检查Vulnerable subject弱势受试者Wash-out period洗脱期Well-being福利,健康。
临床试验英语词汇

缩略语英文全称中文全称ABE Average Bioequivalence 平均生物等效性AC Active control 阳性对照ADE Adverse Drug Event 药物不良事件ADR Adverse Drug Reaction 药物不良反应AE Adverse Event 不良事件AI Assistant Investigator 助理研究者ALB Albumin 白蛋白ALD Approximate Lethal Dose 近似致死剂量ALP Alkaline phosphatase 碱性磷酸酶ALT Alanine aminotransferase 丙氨酸转氨酶ANDA Abbreviated New Drug Application 简化新药申请ANOVA Analysis of variance 方差分析AST Aspartate aminotransferase 天冬氨酸转氨酶ATR Attenuated total reflection 衰减全反射法BA Bioavailability 生物利用度BE Bioequivalence 生物等效性BMI Body Mass Index 体质指数BUN Blood urea nitrogen 血尿素氮CATD Computer-assisted trial design 计算机辅助试验设计CDER Center of Drug Evaluation and Research 药品评价和研究中心CFR Code of Federal Regulation 美国联邦法规CI Co-Investigator 合作研究者CI Confidence Interval 可信区间COI Coordinating Investigator 协调研究者CRC Clinical Research Coordinator 临床研究协调者CRF Case Report/Record Form 病历报告表/病例记录表CRO Contract Research Organization 合同研究组织CSA Clinical Study Application 临床研究申请CTA Clinical Trial Application 临床试验申请CTP Clinical Trial Protocol 临床试验方案CTR Clinical Trial Report 临床试验报告CTX Clinical Trial Exemption 临床试验免责CHMP Committee for Medicinal 人用药委会Products for Human UseDSC Differential scanning 差示扫描热量计DSMB Data Safety and monitoring Board 数据安全及监控委员会EDC Electronic Data Capture 电子数据采集系统EDP Electronic Data Processing 电子数据处理系统EWP Europe Working Party 欧洲工作组FDA Food and Drug Administration 美国食品与药品管理局FR Final Report 总结报告GCP Good Clinical Practice 药物临床试验质量管理规范GCP Good Laboratory Practice 药物非临床试验质量管理规范GLU Glucose 葡萄糖GMP Good Manufacturing Practice 药品生产质量管理规范HEV Health economic evaluation 健康经济学评价IB Investigator’s Brochure 研究者手册IBE IndividualBioequivalence 个体生物等效性IC Informed Consent 知情同意ICF Informed Consent Form 知情同意书ICH International Conference on Harmonization 国际协调会议IDM Independent Data Monitoring 独立数据监察IDMC Independent Data Monitoring Committee 独立数据监察委员会IEC Independent Ethics Committee 独立伦理委员会IND Investigational New Drug 新药临床研究IRB Institutional Review Board 机构审查委员会ITT Intention-to –treat 意向性分析IVD In Vitro Diagnostic 体外诊断IVRS Interactive Voice Response System 互动语音应答系统LD50 Medial lethal dose 半数致死剂量LLOQ Lower Limit of quantitation 定量下限LOCF Last observation carry forward 最接近一次观察的结转LOQ Limit of Quantitation 检测限MA Marketing Approval/Authorization 上市许可证MCA Medicines Control Agency 英国药品监督局MHW Ministry of Health and Welfare 日本卫生福利部MRT Mean residence time 平均滞留时间MTD Maximum Tolerated Dose 最大耐受剂量ND Not detectable 无法定量NDA New Drug Application 新药申请NEC New Drug Entity 新化学实体NIH National Institutes of Health 国家卫生研究所(美国)NMR Nuclear Magnetic Resonance 核磁共振PD Pharmacodynamics 药效动力学PI Principal Investigator 主要研究者PK Pharmacokinetics 药物动力学PL Product License 产品许可证PMA Pre-market Approval (Application) 上市前许可(申请)PP Per protocol 符合方案集PSI Statisticians in the Pharmaceutical Industry 制药业统计学家协会QA Quality Assurance 质量保证QAU Quality Assurance Unit 质量保证部门QC Quality Control 质量控制QWP Quality Working Party 质量工作组RA Regulatory Authorities 监督管理部门REV Revision 修订SA Site Assessment 现场评估SAE Serious Adverse Event 严重不良事件SAP Statistical Analysis Plan 统计分析计划SAR Serious Adverse Reaction 严重不良反应SD Source Data/Document 原始数据/文件SD Subject Diary 受试者日记SDV Source Data Verification 原始数据核准SEL Subject Enrollment Log 受试者入选表SFDA State Food and Drug Administration 国家食品药品监督管理局SI Sponsor-Investigator 申办研究者SI Sub-investigator 助理研究者SIC Subject Identification Code 受试者识别代码SOP Standard Operating Procedure 标准操作规程SPL Study Personnel List 研究人员名单SSL Subject Screening Log 受试者筛选表T&R Test and Reference Product 受试和参比试剂T-BIL Total Bilirubin 总胆红素T-CHO Total Cholesterol 总胆固醇TG Thromboglobulin 血小板球蛋白Tmax Time of maximum concentration 达峰时间TP Total proteinum 总蛋白UAE Unexpected Adverse Event 预料外不良事件WHO World Health Organization 世界卫生组织WHO- WHO International Conference WHO 国际药品管理当局会议ICDR A of Drug Regulatory AuthoritiesAberrant result 异常结果Absorption phase 吸收相Absorption 吸收Accuracy 准确度Accurate 精密度Administer 给药Amendment修正案Approval 批准Assess 估计Audit Report 稽查报告Audit 稽查Auditor 稽查员Analytical run/batch:分析批Benefit 获益Bias 偏性,偏倚Bioequivalence 生物等效Biosimilar /Follow-on biologics 生物仿制药Blank Control 空白对照Blind codes 编制盲底Blind review 盲态检查 /盲态审核Blinding method 盲法Blinding/masking 盲法/设盲Block size 每段的长度Block 层 /分段BCS 生物药剂学分类系统Carryover effect 延滞效应Case history 病历Clinical equivalence 临床等效性Clinical study 临床研究Clinical Trial Report 临床试验报告Comparison 对照Compensation 补偿,赔偿金Compliance 依从性Concomitant 伴随的Conduct 行为Confidence level 置信水平Consistency test 一致性检验Contract/ agreement 协议/合同Control group 对照组Coordinating Committee 协调委员会Crossover design 交叉设计Cross-over Study 交叉研究Cure 痊愈Data management 数据管理Descriptive statistical analysis 描述性统计分析Dichotomies 二分类Dispense 分布Diviation 偏差Documentation 记录/文件Dosage forms 剂型Dose dumping 剂量倾卸(药物迅速释放入血而达到危险浓度)Dose-reaction relation 剂量-反应关系Double blinding 双盲Double dummy 双模Drop out 脱落Effectiveness 疗效Elimination phase 消除相Emergency envelope 应急信件Enantiomers 对映体End point 终点Endpoint criteria/ measurement 终点指标Enterohepatic recycling 肠肝循环Essential Documentation 必需文件Ethical 伦理的Ethics committee 伦理委员会Evaluate 评估Exclusion Criteria 排除标准Excretion 排泄Expedite 促进Extrapolated 外推的Essentially similar product:基本相似药物Factorial design 析因设计Failure 无效,失败Finacing 财务,资金Final point 终点First pass metabolism 首过代谢Fixed-dose procedure 固定剂量法Full analysis set 全分析集GC-FTIR 气相色谱-傅利叶红外联用GC-MS 气相色谱-质谱联用Generic drug 通用名药Gene mutation 基因突变Genotoxicity tests 生殖毒性试验Global assessment variable 全局评价变量Group sequential design 成组序贯设计Hypothesis test 假设检验Highly permeable:高渗透性Highly soluble:高溶解度Highly variable drug:高变异性药物Highly:Variable Drug 高变异性药物HVDP:高变异药物制剂Identification 鉴别,身份证Improvement 好转In vitro 体外In vivo 体内Inclusion Criteria 入选表准Information Gathering 信息收集Initial Meeting 启动会议Inspection 检察/视察Institution Inspection 机构检察Instruction 指令,说明Integrity 完整,正直Intercurrent 中间发生的,间发的Inter-individual variability 个体间变异性Interim analysis 期中分析Investigational Product 试验药物Investigator 研究者Involve 引起,包括IR 红外吸收光谱Innovator Product:原创药Ka 吸收速率常LC-MS 液相色谱-质谱联用logarithmic transformation 对数转换Logic check 逻辑检查Lost of follow up 失访Mask 面具,掩饰Matched pair 匹配配对Metabolism 代谢Missing value 缺失值Mixed effect model 混合效应模式Modified release products 改良释放剂型Monitor 监查员Monitoring Plan 监察计划Monitoring Report 监察报告MS-MS 质谱-质谱联用Multi-center Trial 多中心试验Negative 阴性,否定的Non-clinical Study 非临床研究Non-inferiority 非劣效性Non-Linear Pharmacokinetics 非线性药代动力学Non-parametric statistics 非参数统计方法NTID:窄治疗指数制剂Obedience 依从性Open-blinding 非盲Open-label 非盲Original Medical Record 原始医疗记录Outcome Assessment 结果评价Outcome measurement 结果指标Outlier 离群值OIP 经口服吸收药物Parallel group design 平行组设计Parameter estimation 参数估计Parametric statistics 参数统计方法Patient file 病人档案Patient History 病历Per protocol, PP 符合方案集Permeability 渗透性Pharmacodynamic characteristics 药效学特征Pharmacokinetic characteristics 药代学特征Placebo Control 安慰剂对照Placebo 安慰剂Polytomies 多分类Post-dosing postures 给药后坐姿Potential 潜在的Power 检验效能Precision 精密度Preclinical Study 临床前研究Precursor 母体前体Premature 过早的,早发Primary endpoint 主要终点Primary variable 主要变量Prodrug 药物前体Protocol amendment 方案补正Protocol Amendments 修正案Protocol 试验方案Quality Control Sample:质控样品Rapidly dissolving:快速溶出Racemates 外消旋物Randomization 随机 /随机化Range check 范围检Rating scale 量表Recruit 招募,新会员Replication 可重复Retrieval 取回,补修Revise 修正Risk 风险Run in 准备期Safety evaluation 安全性评价Safety set 安全性评价的数据集Sample Size 样本量、样本大小Sampling schedules 采血计划Scale of ordered categorical ratings 有序分类指标Secondary variable 次要变量Sequence 试验次序Seriousness 严重性Severity 严重程度Significant level 检验水准Simple randomization 简单随机Single Blinding 单盲Site audit 试验机构稽查Solubility 溶解度Specificity 特异性Specify 叙述,说明Sponsor-investigator 申办研究者Standard curve 标准曲线Statistical model 统计模型Statistical tables 统计分析表Steady state 稳态Storage 储存Stratified 分层Study Audit 研究稽查Study Site 研究中心Subgroup 亚组Sub-investigator 助理研究者Subject Enrollment Log 受试者入选表Subject Enrollment 受试者入选Subject Identification Code List 受试者识别代码表Subject Recruitment 受试者招募Subject Screening Log 受试者筛选表Subject 受试者Submit 交付,委托Superiority 检验Supplemental 增补的Supra-bioavailability 超生物利用度(试验药的生物利用度大于对照药)Survival analysis 生存分析System Audit 系统稽查SmPC:药品说明书Standard Sample:标准样品Target variable 目标变量Treatment group 试验组Trial error 试验误差Trial Initial Meeting 试验启动会议Trial Master File 试验总档案Trial Objective 试验目的Trial site 试验场所Triple Blinding 三盲Two one-side test 双单侧检验Therapeutic equivalence:治疗等效性Un-blinding 破盲/揭盲Verify 查证、核实Visual analogy scale 直观类比打分法Vulnerable subject 弱势受试者Wash-out Period 洗脱期Well-being 福利,健康Withdraw 撤回,取消药代动力学参数Ae(0-t):给药到t时尿中排泄的累计原形药。
临床试验常见英语词汇

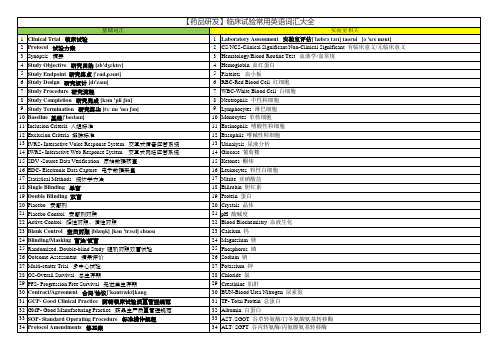
临床试验常见英语词汇基础词汇Clinical Trial 临床试验Protocol 试验方案Synopsis 摘要Study Objective 研究目的Study Endpoint 研究终点Study Design 研究设计Study Procedure 研究流程Study Completion 研究完成Study Termination 研究终止Baseline 基线Inclusion Criteria 入组标准Exclusion Criteria 排除标准IVRS- Interactive Voice Response System 交互式语音应答系统IWRS- Interactive Web Response System 交互式网络应答系统SDV -Source Data Verification 原始数据核查EDC- Electronic Data Capture 电子数据采集Statistical Methods 统计学方法Single Blinding 单盲Double Blinding 双盲Placebo 安慰剂Placebo Control 安慰剂对照Active Control 阳性对照、活性对照Blank Control 空白对照Blinding/Masking 盲法/设盲Randomized, Double-blind Study 随机对照双盲试验Outcome Assessment 结果评价Multi-center Trial 多中心试验OS-Overall Survival 总生存期PFS- Progression Free Survival 无进展生存期Contract/Agreement 合同/协议GCP- Good Clinical Practice 药物临床试验质量管理规范GMP- Good Manufacturing Practice 药品生产质量管理规范SOP- Standard Operating Procedure 标准操作规程Protocol Amendments 修正案CRF- Case Report Form 病历报告表IB- Investigator’s Brochure 研究者手册ICF- Informed Consent Form 知情同意书QA- Quality Assurance 质量保证QC-Quality Control 质量控制Monitoring 监查Monitor 监查员Audit 稽查Auditor 稽查员Inspection 视察CFDA - China Food and Drug Administration 国家食品药品监督管理局FDA- Food and Drug Administration 美国食品与药品监督管理局ICH- International Conference on Harmonization 国际协调会议IEC Independent Ethics Committee 独立伦理委员会IRB-Institutional Review Board 机构审查委员会Sponsor 申办者CRO -Contract Research Organization 合同研究组织SMO-Site Management Organization 现场管理组织Investigational Site 研究中心Investigator 研究者PI- Principal Investigator 主要研究者SI- Sub-investigator 助理研究者CRC- Clinical Research Coordinator 临床研究协调员Initial Meeting 启动会SIV-Site Initiation Visit 中心启动访视。
临床试验英语词汇

专业术语、缩略语中英对照表缩略语英文全称中文全称ABE Average Bioequivalence 平均生物等效性AC Active control 阳性对照ADE Adverse Drug Event 药物不良事件ADR Adverse Drug Reaction 药物不良反应AE Adverse Event 不良事件AI Assistant Investigator 助理研究者ALB Albumin 白蛋白ALD Approximate Lethal Dose 近似致死剂量ALP Alkaline phosphatase 碱性磷酸酶ALT Alanine aminotransferase 丙氨酸转氨酶ANDA Abbreviated New Drug Application 简化新药申请ANOVA Analysis of variance 方差分析AST Aspartate aminotransferase 天冬氨酸转氨酶ATR Attenuated total reflection 衰减全反射法BA Bioavailability 生物利用度BE Bioequivalence 生物等效性BMI Body Mass Index 体质指数BUN Blood urea nitrogen 血尿素氮CATD Computer-assisted trial design 计算机辅助试验设计CDER Center of Drug Evaluation and Research 药品评价和研究中心CFR Code of Federal Regulation 美国联邦法规CI Co-Investigator 合作研究者CI Confidence Interval 可信区间COI Coordinating Investigator 协调研究者CRC Clinical Research Coordinator 临床研究协调者CRF Case Report/Record Form 病历报告表/病例记录表CRO Contract Research Organization 合同研究组织CSA Clinical Study Application 临床研究申请CTA Clinical Trial Application 临床试验申请CTP Clinical Trial Protocol 临床试验方案CTR Clinical Trial Report 临床试验报告CTX Clinical Trial Exemption 临床试验免责CHMP Committee for Medicinal 人用药委会Products for Human UseDSC Differential scanning 差示扫描热量计DSMB Data Safety and monitoring Board 数据安全及监控委员会EDC Electronic Data Capture 电子数据采集系统EDP Electronic Data Processing 电子数据处理系统EWP Europe Working Party 欧洲工作组FDA Food and Drug Administration 美国食品与药品管理局FR Final Report 总结报告GCP Good Clinical Practice 药物临床试验质量管理规GCP Good Laboratory Practice 药物非临床试验质量管理规GLU Glucose 葡萄糖GMP Good Manufacturing Practice 药品生产质量管理规HEV Health economic evaluation 健康经济学评价IB Investigator’s Brochure 研究者手册IBE IndividualBioequivalence 个体生物等效性IC Informed Consent 知情同意ICF Informed Consent Form 知情同意书ICH International Conference on Harmonization 国际协调会议IDM Independent Data Monitoring 独立数据监察IDMC Independent Data Monitoring Committee 独立数据监察委员会IEC Independent Ethics Committee 独立伦理委员会IND Investigational New Drug 新药临床研究IRB Institutional Review Board 机构审查委员会ITT Intention-to –treat 意向性分析IVD In Vitro Diagnostic 体外诊断IVRS Interactive Voice Response System 互动语音应答系统LD50 Medial lethal dose 半数致死剂量LLOQ Lower Limit of quantitation 定量下限LOCF Last observation carry forward 最接近一次观察的结转LOQ Limit of Quantitation 检测限MA Marketing Approval/Authorization 上市许可证MCA Medicines Control Agency 英国药品监督局MHW Ministry of Health and Welfare 日本卫生福利部MRT Mean residence time 平均滞留时间MTD Maximum Tolerated Dose 最大耐受剂量ND Not detectable 无法定量NDA New Drug Application 新药申请NEC New Drug Entity 新化学实体NIH National Institutes of Health 国家卫生研究所(美国)NMR Nuclear Magnetic Resonance 核磁共振PD Pharmacodynamics 药效动力学PI Principal Investigator 主要研究者PK Pharmacokinetics 药物动力学PL Product License 产品许可证PMA Pre-market Approval (Application) 上市前许可(申请)PP Per protocol 符合方案集PSI Statisticians in the Pharmaceutical Industry 制药业统计学家协会QA Quality Assurance 质量保证QAU Quality Assurance Unit 质量保证部门QC Quality Control 质量控制QWP Quality Working Party 质量工作组RA Regulatory Authorities 监督管理部门REV Revision 修订SA Site Assessment 现场评估SAE Serious Adverse Event 严重不良事件SAP Statistical Analysis Plan 统计分析计划SAR Serious Adverse Reaction 严重不良反应SD Source Data/Document 原始数据/文件SD Subject Diary 受试者日记SDV Source Data Verification 原始数据核准SEL Subject Enrollment Log 受试者入选表SFDA State Food and Drug Administration 国家食品药品监督管理局SI Sponsor-Investigator 申办研究者SI Sub-investigator 助理研究者SIC Subject Identification Code 受试者识别代码SOP Standard Operating Procedure 标准操作规程SPL Study Personnel List 研究人员SSL Subject Screening Log 受试者筛选表T&R Test and Reference Product 受试和参比试剂T-BIL Total Bilirubin 总胆红素T-CHO Total Cholesterol 总胆固醇TG Thromboglobulin 血小板球蛋白Tmax Time of maximum concentration 达峰时间TP Total proteinum 总蛋白UAE Unexpected Adverse Event 预料外不良事件WHO World Health Organization 世界卫生组织WHO- WHO International Conference WHO 国际药品管理当局会议ICDR A of Drug Regulatory AuthoritiesAberrant result 异常结果Absorption phase 吸收相Absorption 吸收Accuracy 准确度Accurate 精密度Administer 给药Amendment修正案Approval 批准Assess 估计Audit Report 稽查报告Audit 稽查Auditor 稽查员Analytical run/batch:分析批Benefit 获益Bias 偏性,偏倚Bioequivalence 生物等效Biosimilar /Follow-on biologics 生物仿制药Blank Control 空白对照Blind codes 编制盲底Blind review 盲态检查/盲态审核Blinding method 盲法Blinding/masking 盲法/设盲Block size 每段的长度Block 层/分段BCS 生物药剂学分类系统Carryover effect 延滞效应Case history 病历Clinical equivalence 临床等效性Clinical study 临床研究Clinical Trial Report 临床试验报告Comparison 对照Compensation 补偿,赔偿金Compliance 依从性Concomitant 伴随的Conduct 行为Confidence level 置信水平Consistency test 一致性检验Contract/ agreement 协议/合同Control group 对照组Coordinating Committee 协调委员会Crossover design 交叉设计Cross-over Study 交叉研究Cure 痊愈Data management 数据管理Descriptive statistical analysis 描述性统计分析Dichotomies 二分类Dispense 分布Diviation 偏差Documentation 记录/文件Dosage forms 剂型Dose dumping 剂量倾卸(药物迅速释放入血而达到危险浓度)Dose-reaction relation 剂量-反应关系Double blinding 双盲Double dummy 双模Drop out 脱落Effectiveness 疗效Elimination phase 消除相Emergency envelope 应急信件Enantiomers 对映体End point 终点Endpoint criteria/ measurement 终点指标Enterohepatic recycling 肠肝循环Essential Documentation 必需文件Ethical 伦理的Ethics committee 伦理委员会Evaluate 评估Exclusion Criteria 排除标准Excretion 排泄Expedite 促进Extrapolated 外推的Essentially similar product:基本相似药物Factorial design 析因设计Failure 无效,失败Finacing 财务,资金Final point 终点First pass metabolism 首过代Fixed-dose procedure 固定剂量法Full analysis set 全分析集GC-FTIR 气相色谱-傅利叶红外联用GC-MS 气相色谱-质谱联用Generic drug 通用名药Gene mutation 基因突变Genotoxicity tests 生殖毒性试验Global assessment variable 全局评价变量Group sequential design 成组序贯设计Hypothesis test 假设检验Highly permeable:高渗透性Highly soluble:高溶解度Highly variable drug:高变异性药物Highly:Variable Drug 高变异性药物HVDP:高变异药物制剂Identification 鉴别,Improvement 好转In vitro 体外In vivo 体Inclusion Criteria 入选表准Information Gathering 信息收集Initial Meeting 启动会议Inspection 检察/视察Institution Inspection 机构检察Instruction 指令,说明Integrity 完整,正直Intercurrent 中间发生的,间发的Inter-individual variability 个体间变异性Interim analysis 期中分析Investigational Product 试验药物Investigator 研究者Involve 引起,包括IR 红外吸收光谱Innovator Product:原创药Ka 吸收速率常LC-MS 液相色谱-质谱联用logarithmic transformation 对数转换Logic check 逻辑检查Lost of follow up 失访Mask 面具,掩饰Matched pair 匹配配对Metabolism 代Missing value 缺失值Mixed effect model 混合效应模式Modified release products 改良释放剂型Monitor 监查员Monitoring Plan 监察计划Monitoring Report 监察报告MS-MS 质谱-质谱联用Multi-center Trial 多中心试验Negative 阴性,否定的Non-clinical Study 非临床研究Non-inferiority 非劣效性Non-Linear Pharmacokinetics 非线性药代动力学Non-parametric statistics 非参数统计方法NTID:窄治疗指数制剂Obedience 依从性Open-blinding 非盲Open-label 非盲Original Medical Record 原始医疗记录Outcome Assessment 结果评价Outcome measurement 结果指标Outlier 离群值OIP 经口服吸收药物Parallel group design 平行组设计Parameter estimation 参数估计Parametric statistics 参数统计方法Patient file 病人档案Patient History 病历Per protocol,PP 符合方案集Permeability 渗透性Pharmacodynamic characteristics 药效学特征Pharmacokinetic characteristics 药代学特征Placebo Control 安慰剂对照Placebo 安慰剂Polytomies 多分类Post-dosing postures 给药后坐姿Potential 潜在的Power 检验效能Precision 精密度Preclinical Study 临床前研究Precursor 母体前体Premature 过早的,早发Primary endpoint 主要终点Primary variable 主要变量Prodrug 药物前体Protocol amendment 方案补正Protocol Amendments 修正案Protocol 试验方案Quality Control Sample:质控样品Rapidly dissolving:快速溶出Racemates 外消旋物Randomization 随机/随机化Range check 围检Rating scale 量表Recruit 招募,新会员Replication 可重复Retrieval 取回,补修Revise 修正Risk 风险Run in 准备期Safety evaluation 安全性评价Safety set 安全性评价的数据集Sample Size 样本量、样本大小Sampling schedules 采血计划Scale of ordered categorical ratings 有序分类指标Secondary variable 次要变量Sequence 试验次序Seriousness 严重性Severity 严重程度Significant level 检验水准Simple randomization 简单随机Single Blinding 单盲Site audit 试验机构稽查Solubility 溶解度Specificity 特异性Specify 叙述,说明Sponsor-investigator 申办研究者Standard curve 标准曲线Statistical model 统计模型Statistical tables 统计分析表Steady state 稳态Storage 储存Stratified 分层Study Audit 研究稽查Study Site 研究中心Subgroup 亚组Sub-investigator 助理研究者Subject Enrollment Log 受试者入选表Subject Enrollment 受试者入选Subject Identification Code List 受试者识别代码表Subject Recruitment 受试者招募Subject Screening Log 受试者筛选表Subject 受试者Submit 交付,委托Superiority 检验Supplemental 增补的Supra-bioavailability 超生物利用度(试验药的生物利用度大于对照药)Survival analysis 生存分析System Audit 系统稽查SmPC:药品说明书Standard Sample:标准样品Target variable 目标变量Treatment group 试验组Trial error 试验误差Trial Initial Meeting 试验启动会议Trial Master File 试验总档案Trial Objective 试验目的Trial site 试验场所Triple Blinding 三盲Two one-side test 双单侧检验Therapeutic equivalence:治疗等效性Un-blinding 破盲/揭盲Verify 查证、核实Visual analogy scale 直观类比打分法Vulnerable subject 弱势受试者Wash-out Period 洗脱期Well-being 福利,健康Withdraw 撤回,取消药代动力学参数Ae(0-t):给药到t时尿中排泄的累计原形药。
- 1、下载文档前请自行甄别文档内容的完整性,平台不提供额外的编辑、内容补充、找答案等附加服务。
- 2、"仅部分预览"的文档,不可在线预览部分如存在完整性等问题,可反馈申请退款(可完整预览的文档不适用该条件!)。
- 3、如文档侵犯您的权益,请联系客服反馈,我们会尽快为您处理(人工客服工作时间:9:00-18:30)。
专业术语、缩略语中英对照表缩略语英文全称中文全称ABE Average Bioequivalence 平均生物等效性AC Active control 阳性对照ADE Adverse Drug Event 药物不良事件ADR Adverse Drug Reaction 药物不良反应AE Adverse Event 不良事件AI Assistant Investigator 助理研究者ALB Albumin 白蛋白ALD Approximate Lethal Dose 近似致死剂量ALP Alkaline phosphatase 碱性磷酸酶ALT Alanine aminotransferase 丙氨酸转氨酶ANDA Abbreviated New Drug Application 简化新药申请ANOVA Analysis of variance 方差分析AST Aspartate aminotransferase 天冬氨酸转氨酶ATR Attenuated total reflection 衰减全反射法BA Bioavailability 生物利用度BE Bioequivalence 生物等效性BMI Body Mass Index 体质指数BUN Blood urea nitrogen 血尿素氮CATD Computer-assisted trial design 计算机辅助试验设计CDER Center of Drug Evaluation and Research 药品评价和研究中心CFR Code of Federal Regulation 美国联邦法规CI Co-Investigator 合作研究者CI Confidence Interval 可信区间COI Coordinating Investigator 协调研究者CRC Clinical Research Coordinator 临床研究协调者CRF Case Report/Record Form 病历报告表/病例记录表CRO Contract Research Organization 合同研究组织CSA Clinical Study Application 临床研究申请CTA Clinical Trial Application 临床试验申请CTP Clinical Trial Protocol 临床试验方案CTR Clinical Trial Report 临床试验报告CTX Clinical Trial Exemption 临床试验免责CHMP Committee for Medicinal 人用药委会Products for Human UseDSC Differential scanning 差示扫描热量计DSMB Data Safety and monitoring Board 数据安全及监控委员会EDC Electronic Data Capture 电子数据采集系统EDP Electronic Data Processing 电子数据处理系统EWP Europe Working Party 欧洲工作组FDA Food and Drug Administration 美国食品与药品管理局FR Final Report 总结报告GCP Good Clinical Practice 药物临床试验质量管理规范GCP Good Laboratory Practice 药物非临床试验质量管理规范GLU Glucose 葡萄糖GMP Good Manufacturing Practice 药品生产质量管理规范HEV Health economic evaluation 健康经济学评价IB Investigator’s Brochure研究者手册IBE IndividualBioequivalence 个体生物等效性IC Informed Consent 知情同意ICF Informed Consent Form 知情同意书ICH International Conference on Harmonization 国际协调会议IDM Independent Data Monitoring 独立数据监察IDMC Independent Data Monitoring Committee 独立数据监察委员会IEC Independent Ethics Committee 独立伦理委员会IND Investigational New Drug 新药临床研究IRB Institutional Review Board 机构审查委员会ITT Intention-to –treat 意向性分析IVD In Vitro Diagnostic 体外诊断IVRS Interactive Voice Response System 互动语音应答系统LD50 Medial lethal dose 半数致死剂量LLOQ Lower Limit of quantitation 定量下限LOCF Last observation carry forward 最接近一次观察的结转LOQ Limit of Quantitation 检测限MA Marketing Approval/Authorization 上市许可证MCA Medicines Control Agency 英国药品监督局MHW Ministry of Health and Welfare 日本卫生福利部MRT Mean residence time 平均滞留时间MTD Maximum Tolerated Dose 最大耐受剂量ND Not detectable 无法定量NDA New Drug Application 新药申请NEC New Drug Entity 新化学实体NIH National Institutes of Health 国家卫生研究所(美国)NMR Nuclear Magnetic Resonance 核磁共振PD Pharmacodynamics 药效动力学PI Principal Investigator 主要研究者PK Pharmacokinetics 药物动力学PL Product License 产品许可证PMA Pre-market Approval (Application) 上市前许可(申请)PP Per protocol 符合方案集PSI Statisticians in the Pharmaceutical Industry 制药业统计学家协会QA Quality Assurance 质量保证QAU Quality Assurance Unit 质量保证部门QC Quality Control 质量控制QWP Quality Working Party 质量工作组RA Regulatory Authorities 监督管理部门REV Revision 修订SA Site Assessment 现场评估SAE Serious Adverse Event 严重不良事件SAP Statistical Analysis Plan 统计分析计划SAR Serious Adverse Reaction 严重不良反应SD Source Data/Document 原始数据/文件SD Subject Diary 受试者日记SDV Source Data Verification 原始数据核准SEL Subject Enrollment Log 受试者入选表SFDA State Food and Drug Administration 国家食品药品监督管理局SI Sponsor-Investigator 申办研究者SI Sub-investigator 助理研究者SIC Subject Identification Code 受试者识别代码SOP Standard Operating Procedure 标准操作规程SPL Study Personnel List 研究人员名单SSL Subject Screening Log 受试者筛选表T&R Test and Reference Product 受试和参比试剂T-BIL Total Bilirubin 总胆红素T-CHO Total Cholesterol 总胆固醇TG Thromboglobulin 血小板球蛋白Tmax Time of maximum concentration 达峰时间TP Total proteinum 总蛋白UAE Unexpected Adverse Event 预料外不良事件WHO World Health Organization 世界卫生组织WHO- WHO International Conference WHO 国际药品管理当局会议ICDR A of Drug Regulatory AuthoritiesAberrant result 异常结果Absorption phase 吸收相Absorption 吸收Accuracy 准确度Accurate 精密度Administer 给药Amendment修正案Approval 批准Assess 估计Audit Report 稽查报告Audit 稽查Auditor 稽查员Analytical run/batch:分析批Benefit 获益Bias 偏性,偏倚Bioequivalence 生物等效Biosimilar /Follow-on biologics 生物仿制药Blank Control 空白对照Blind codes 编制盲底Blind review 盲态检查/盲态审核Blinding method 盲法Blinding/masking 盲法/设盲Block size 每段的长度Block 层/分段BCS 生物药剂学分类系统Carryover effect 延滞效应Case history 病历Clinical equivalence 临床等效性Clinical study 临床研究Clinical Trial Report 临床试验报告Comparison 对照Compensation 补偿,赔偿金Compliance 依从性Concomitant 伴随的Conduct 行为Confidence level 置信水平Consistency test 一致性检验Contract/ agreement 协议/合同Control group 对照组Coordinating Committee 协调委员会Crossover design 交叉设计Cross-over Study 交叉研究Cure 痊愈Data management 数据管理Descriptive statistical analysis 描述性统计分析Dichotomies 二分类Dispense 分布Diviation 偏差Documentation 记录/文件Dosage forms 剂型Dose dumping 剂量倾卸(药物迅速释放入血而达到危险浓度)Dose-reaction relation 剂量-反应关系Double blinding 双盲Double dummy 双模Drop out 脱落Effectiveness 疗效Elimination phase 消除相Emergency envelope 应急信件Enantiomers 对映体End point 终点Endpoint criteria/ measurement 终点指标Enterohepatic recycling 肠肝循环Essential Documentation 必需文件Ethical 伦理的Ethics committee 伦理委员会Evaluate 评估Exclusion Criteria 排除标准Excretion 排泄Expedite 促进Extrapolated 外推的Essentially similar product:基本相似药物Factorial design 析因设计Failure 无效,失败Finacing 财务,资金Final point 终点First pass metabolism 首过代谢Fixed-dose procedure 固定剂量法Full analysis set 全分析集GC-FTIR 气相色谱-傅利叶红外联用GC-MS 气相色谱-质谱联用Generic drug 通用名药Gene mutation 基因突变Genotoxicity tests 生殖毒性试验Global assessment variable 全局评价变量Group sequential design 成组序贯设计Hypothesis test 假设检验Highly permeable:高渗透性Highly soluble:高溶解度Highly variable drug:高变异性药物Highly:Variable Drug 高变异性药物HVDP:高变异药物制剂Identification 鉴别,身份证Improvement 好转In vitro 体外In vivo 体内Inclusion Criteria 入选表准Information Gathering 信息收集Initial Meeting 启动会议Inspection 检察/视察Institution Inspection 机构检察Instruction 指令,说明Integrity 完整,正直Intercurrent 中间发生的,间发的Inter-individual variability 个体间变异性Interim analysis 期中分析Investigational Product 试验药物Investigator 研究者Involve 引起,包括IR 红外吸收光谱Innovator Product:原创药Ka 吸收速率常LC-MS 液相色谱-质谱联用logarithmic transformation 对数转换Logic check 逻辑检查Lost of follow up 失访Mask 面具,掩饰Matched pair 匹配配对Metabolism 代谢Missing value 缺失值Mixed effect model 混合效应模式Modified release products 改良释放剂型Monitor 监查员Monitoring Plan 监察计划Monitoring Report 监察报告MS-MS 质谱-质谱联用Multi-center Trial 多中心试验Negative 阴性,否定的Non-clinical Study 非临床研究Non-inferiority 非劣效性Non-Linear Pharmacokinetics 非线性药代动力学Non-parametric statistics 非参数统计方法NTID:窄治疗指数制剂Obedience 依从性Open-blinding 非盲Open-label 非盲Original Medical Record 原始医疗记录Outcome Assessment 结果评价Outcome measurement 结果指标Outlier 离群值OIP 经口服吸收药物Parallel group design 平行组设计Parameter estimation 参数估计Parametric statistics 参数统计方法Patient file 病人档案Patient History 病历Per protocol,PP 符合方案集Permeability 渗透性Pharmacodynamic characteristics 药效学特征Pharmacokinetic characteristics 药代学特征Placebo Control 安慰剂对照Placebo 安慰剂Polytomies 多分类Post-dosing postures 给药后坐姿Potential 潜在的Power 检验效能Precision 精密度Preclinical Study 临床前研究Precursor 母体前体Premature 过早的,早发Primary endpoint 主要终点Primary variable 主要变量Prodrug 药物前体Protocol amendment 方案补正Protocol Amendments 修正案Protocol 试验方案Quality Control Sample:质控样品Rapidly dissolving:快速溶出Racemates 外消旋物Randomization 随机/随机化Range check 范围检Rating scale 量表Recruit 招募,新会员Replication 可重复Retrieval 取回,补修Revise 修正Risk 风险Run in 准备期Safety evaluation 安全性评价Safety set 安全性评价的数据集Sample Size 样本量、样本大小Sampling schedules 采血计划Scale of ordered categorical ratings 有序分类指标Secondary variable 次要变量Sequence 试验次序Seriousness 严重性Severity 严重程度Significant level 检验水准Simple randomization 简单随机Single Blinding 单盲Site audit 试验机构稽查Solubility 溶解度Specificity 特异性Specify 叙述,说明Sponsor-investigator 申办研究者Standard curve 标准曲线Statistical model 统计模型Statistical tables 统计分析表Steady state 稳态Storage 储存Stratified 分层Study Audit 研究稽查Study Site 研究中心Subgroup 亚组Sub-investigator 助理研究者Subject Enrollment Log 受试者入选表Subject Enrollment 受试者入选Subject Identification Code List 受试者识别代码表Subject Recruitment 受试者招募Subject Screening Log 受试者筛选表Subject 受试者Submit 交付,委托Superiority 检验Supplemental 增补的Supra-bioavailability 超生物利用度(试验药的生物利用度大于对照药)Survival analysis 生存分析System Audit 系统稽查SmPC:药品说明书Standard Sample:标准样品Target variable 目标变量Treatment group 试验组Trial error 试验误差Trial Initial Meeting 试验启动会议Trial Master File 试验总档案Trial Objective 试验目的Trial site 试验场所Triple Blinding 三盲Two one-side test 双单侧检验Therapeutic equivalence:治疗等效性Un-blinding 破盲/揭盲Verify 查证、核实Visual analogy scale 直观类比打分法Vulnerable subject 弱势受试者Wash-out Period 洗脱期Well-being 福利,健康Withdraw 撤回,取消药代动力学参数Ae(0-t):给药到t时尿中排泄的累计原形药。
