《量子化学》期末考试复习题
量子化学课程习题及标准答案

量子化学习题及标准答案Chapter 011. A certain one-particle, one-dimensional system has/2bmx ibt e ae --=ψ, where a and b are constants and m is the particle ’s mass. Find the potential-energy function V for this system. (Hint : Use the time-dependent Schrodinger equation.)Solution :As ψ(x,t) is known, we can derive the corresponding derivatives.⎪⎪⎩⎪⎪⎨⎧ψ+ψ-=∂ψ∂ψ-=∂ψ∂⇒=ψ--222222/42),(),(),(2 x m b bm xt x ib t t x e ae t x bmx ibtAccording to time-dependent Schroedinger equation,substituting into the derivatives, we get222),(mx b t x V =2. At a certain instant of time, a one-particle, one-dimensional system has bx xe b /||2/13)/2(-=ψ, where b = 3.000 nm. If a measurement of x is made at this time in the system, find the probability that the result (a) lies between 0.9000 nm and 0.9001 nm (treat this interval as infinitesimal); (b) lies between 0 and 2 nm (use the table of integrals, if necessary). (c) For what value of x is the probability density a minimum? (There is no need to use calculus to answer this.) (d) Verify that ψ is normalized.Solution :a) The probability of finding an particle in a space between x and x+dx is given by6/223210*29.32--==ψ=dx e x b dx P b x b) 0753.02910*20/223==⎰--dx e x bP b x c) Clearly, the minimum of probability density is at x=0, where the probability densityvanishes. d)4220/223/223/2232===ψ=⎰⎰⎰⎰+∞-+∞∞--+∞∞--+∞∞-d e x b dx e x b dx e x b dx P b x b x b x3. A one-particle, one-dimensional system has the state function2222/4/16/4/12)/32)((cos )/2)((sin c x c x xe c at e c at --+=ψππ where a is a constant and c = 2.000 Å. If the particle ’s position is measured at t = 0, estimate the probability that the result will lie between 2.000 Å and 2.001 Å.Solution :when t=0, the wavefunction is simplified as441610*158.2)32(),(22--==ψc x xe c t x πChapter 021. Consider an electron in a one-dimensional box of length2.000Å with the left end of the box at x = 0. (a) Suppose we have one million of these systems, each in the n = 1 state, and we measure the x coordinate of the electron in each system. About how many times will the electron be found between 0.600 Å and 0.601 Å? Consider the interval to be infinitesimal. Hint: Check whether your calculator is set to degrees or radians. (b) Suppose we have a large number of these systems, each in the n =1 state, and we measure the x coordinate of the electron in each system and find the electron between 0.700 Å and 0.701 Å in 126 of the measurements. In about how many measurements will the electron be found between 1.000 Å and 1.001 Å?Solution: a) In a 1D box, the energy and wave-function of a micro-system are given by)sin(2,22222x ln l ml n E πψπ== therefore, the probability density of finding the electron between 0.600 and 0.601 Å is65510*545.6)(sin 242⇒==-dx x ln l P πb) From the definition of probability, the probability of finding an electron between x and x+dx is given bydx x l n l P )(sin 22π= As the number of measurements of finding the electron between 0.700 and 0.701 Å is known, the number of system is1(sin 22*158712158712001.0)7.02*1(sin 2212612622=⇒===πP P N2. When a particle of mass 9.1*10-28 g in acertain one-dimensional box goes from the n = 5 level to the n = 2 level, it emits a photon offrequency 6.0*1014 s -1. Find the length of thebox.Solution.lml h n n ml n n E lowerup lowerup 36222222222110*26646.18)(2)(-=-=-=∆ π3. An electron in a stationary state of a one-dimensional box of length 0.300 nm emitsa photon of frequency 5.05*1015 s -1. Find theinitial and final quantum numbers for this transition.Solution:2,388)(2)(22222222222===-⇒=-=-=∆lower upper lower up lower up lower up n n n n hv ml h n n ml n n E π4. For the particle in a one-dimensional box of length l , we could have put the coordinate origin at the center of the box. Find the wave functions and energy levels for this choice of origin.Solution: The wavefunction for a particle in a one-dimernsional box can be written as)2()2()(x mE BSin x mE ACos x+=ψ If the coordinate origin is defined at the center of the box, the boundary conditions are given as2()22(0)(2()22(0)(22mE BSin l mE ACos x mE BSin l mE ACos x l x lx +⇒=-⇒==-= ψψ Combining Eq1 with Eq2, we get)4(,0)22()3(,0)22(Eq l mE BSin Eq l mE ACos == Eq3 leads to A=0, or )22(l mE Cos =0. We willdiscuss both situations in the following section.If A=0, B must be non-zero number otherwise the wavefunction vanishes.2220)22(02mlh n E n l mE l mE Sin B π=⇒=⇒=⇒≠If A ≠08)12()21(220)22(00)22(0)22(0222mlh n E n l mE l mE Cos B l mE Sin l mE Cos A ψπ⇒+=⇒+=⇒==⇒≠⇒=⇒≠5. For an electron in a certain rectangular well with a depth of 20.0 eV , the lowest energy lies 3.00 eV above the bottom of the well. Find the width of this well. Hint : Use tanθ = sin θ/cos θ Solution : For the particle in a certain rectangular well, the E fulfill with )2sin()2()2cos()(21010l mE V E l mE E E V ---=- Substituting into the V and E, we get1010011110*64.22)7954.0(7954.022)(2)2()2()2(-----=⇒+-=⇒+-=⇒-=--==lowest l mE n l n l mE V E E E V l mE Tan l mE Cos l mE Sin ππChapter 03 1. If A ˆf (x ) = 3x 2 f (x ) + 2xd f /dx , give an expression for Aˆ. Solution :Extracting f(x) from the known equation leads to the expression of Adx d x x A 23ˆ2+=2. (a) Show that (Aˆ+Bˆ)2 = (Bˆ+Aˆ)2for any twooperators. (b) Under what conditions is (Aˆ+B ˆ)2 equal to A ˆ2+2A ˆB ˆ+B ˆ2?Solution: a)2222ˆ()ˆˆ)(ˆˆ(ˆˆˆˆˆˆˆˆˆˆˆ)ˆˆ)(ˆˆ()ˆˆ(B A B A BA B A A B B A B B AA B A B A B A =++=+++=+++=++=+ b) BA AB A B B A A B A ˆˆ2ˆˆˆˆˆˆˆ)ˆˆ(2222++=+++=+If and only if A and B commute, (Aˆ+B ˆ)2 equals to A ˆ2+2A ˆB ˆ+B ˆ23. If A ˆ = d 2/dx 2 and B ˆ = x 2, find (a) A ˆB ˆx 3; (b)B ˆA ˆx 3; (c) A ˆB ˆf (x ); (d)B ˆA ˆf (x )Solution: a)3522320ˆˆxx dxd x B A == b)3322236ˆˆx x dxd x x A B == c))()(4)(2)]()(2[)]([)(ˆˆ2222222x f dxd x x f dx d x x f x f dxd x x xf dx d x f x dx d x f B A ++=+==d))()()(ˆˆ222222x f dxd x x f dx d x x f A B ==4. Classify these operators as linear ornonlinear: (a) 3x 2d 2/dx 2; (b) ( )2; (c) ∫ dx ; (d) exp; (e) ∑=n x 1.Solution:Linear operator is subject to the following condition.fA c cf A g A f A g f A ˆ)(ˆˆˆ)(ˆ=+=+ a) Linearb) Nonlinear c) Linear d) Nonlinear e) Linear5. The Laplace transform operator Lˆ isdefined by⎰∞-=0)()(ˆdx x f e x f L px(a) Is L ˆ linear? (b) EvaluateLˆ(1). (c)Evaluate L ˆe ax , assuming that p >a .Solution:a) L is a linear operator b)0,1)1(ˆ0>==⎰∞-p p dx e L px c)pdx e dx e e e L x a p ax px ax ===⎰⎰∞--∞-)(ˆ0)(06. We define the translation operator hT ˆ by hT ˆf (x ) = f (x + h ). (a) Is hT ˆ a linear operator? (b)Evaluate (2ˆ3ˆ121+-T T )x 2.Solution:a) The translation operator is linear operator b)2212212121(2ˆ3ˆ)2ˆ3ˆ(x x x T x T x T T +=+-=+-7. Evaluate the commutators (a) [x ˆ,xpˆ]; (b) [x ˆ,2ˆxp]; (c) [x ˆ,yp ˆ]; (d) [x ˆ, ),,(ˆz y x V ]; (e) [x ˆ,H ˆ]; (f) [z y xˆˆˆ, 2ˆxp ]. Solution: a)i xx x x i x x i p xx =∂∂--∂∂-=∂∂-=)ˆ1ˆ(],ˆ[]ˆ,ˆ[b)xp i p p x p x p p xx x x x x x∂∂==+=222ˆ2)ˆ]ˆ,ˆ[]ˆ,ˆ[ˆ(]ˆ,ˆ[c)0)ˆˆ(],ˆ[]ˆ,ˆ[=∂∂-∂∂-=∂∂-=yx y x i y x i p xyd)0ˆ),,(ˆ),,(ˆˆ)],,(ˆ,ˆ[=-=x z y x V z y x V x z y x V xe)mxT x V T x H x ==+=21,ˆ[]ˆ,ˆ[]ˆˆ,ˆ[]ˆ,ˆ[f)xz y p z y xx∂∂=ˆˆ2]ˆ,ˆˆˆ[22Chapter 041:The one-dimensional harmonic-oscillator is at its first excited state and its wavefunction is given as)21exp()()(2)(24/14/31x x x βπβψ-=please evaluate the expectation values(average values) of kinetic energy (T), potential energy (V) and the total energy.Answer: 1) First of all, check the normalization property of the wavefunction.2) Evaluate the expectation value of kinetic energy.3) Evaluate the expectation value ofpotential energy4) Total Energy = T + V2. The one-dimensional harmonic-oscillator Hamiltonian is2222ˆ22ˆˆxm v mp H xπ+= The raising and lowering operators for thisproblem are defined as]ˆ2ˆ[)2(1ˆ2/1x ivm p m A x π+=+, ]ˆ2ˆ[)2(1ˆ2/1x ivm p m A x π-=-Show thathv H A A 21ˆˆˆ-=-+,hv H A A 21ˆˆˆ+=+-, hv A A-=-+]ˆ,ˆ[ ++=A hv A H ˆ]ˆ,ˆ[, ---=A hv A Hˆ]ˆ,ˆ[ Show that +Aˆ and -A ˆ are indeed ladder operators and that the eigenvalues are spaced at intervals of hv . Since both the kinetic energy and the potential energy are nonnegative, we expect the energy eigenvalues to be nonnegative. Hence there must be a state of minimum energy. Operate on the wavefunction for this state first with -Aˆ and then with +Aˆ and show that the lowest energy eigenvalue ishv 21. Finally, conclude that hvn E )21(+=, n = 0, 1, 2, …Answer:1) Write down the definition of operatordx di px -=ˆ2) Expand the operators in full form.]2[21]2[2122ˆ222222vmxi dx di mA vmxi dx d i mA mx v dx d m H πππ--=+-=+-=-+ 3) Evaluate the corresponding combination ofoperators4242[21]22][2[21ˆ4222[21]]2[2]2[2[21]2[21]22[ˆ21ˆ2122]4222[21]]2[2]2[[21]2[21]2[2121ˆ2122]42[21]4222[21]]2[2]2[[21]2[21]2[21322222222333222222332222222333222222222222222222222222222222222222222222222v dx d ix v dx d i mx v mxi v dxd m i m mx v dx d m vmxi dx d i mH A v dx d i mx v dx d ix v dx d i v dxd m i m vmxi dx d i mx v vmxi dx d i dxd m m vmxi dx d i mmx v dx d m A H hv H hv mx v dx d m x m v dxd vmx dx d x vm vm dx d m vmxi dxd i vmxi vmxi dx d i dx d i m vmxi dx d i mvmxi dx d i m A A hv H hv mx v dx d m x m v vm dxd m x m v dxd vmx dx d x vm vm dx d m vmxi dxd i vmxi vmxi dx d i dx d i m vmxi dx d i mvmxi dx d i m A A πππππππππππππππππππππππππππππππππππππ+---=+-+-=+---=+-++--=+-+-=+=++-=+-++-=+--+--=+---=-=-+-=+--=++---=--+---=--+-=+++--++++=+-=+-=+-=-hvA vmxi m dx d i m hv vmxi m hv dxd i m hv mxi v dx d i v m H A A H ]22121[]221[]21[]42[21ˆˆ222ππππIn the same manner, we can get---=-hvA H A A H ˆˆ 4) Substituting the above communicators into the Schroeidnger equation, we getψψψψψψψψψψψψ------++++++-=-=-=+=+=+==A hv E hvA E A hvA H A A H A hv E hvA E A hvA H A A H E H)(]ˆ[ˆ)(]ˆ[ˆˆThis shows that +Aˆ and -A ˆ are indeed ladder operators and that the eigenvalues are spaced at intervals of hv .5) Suppose that ψ is the eigenfunction with the lowest eigenvalue. ψψlowest E H =ˆAccording to the definition of A_ operator, we haveψψ---=A hv E A H )(ˆAs ψ is the eigenfunction with the lowest eigenvalue, the above equation is fulfilled if and only if 0=-ψAOperating on the wave function for this statefirst with -A ˆ and then with +A ˆ leads to ψψψψhv H hv H A A 21ˆ]21ˆ[0=⇒-==-+Therefore, the lowest energy is 1/2 hv.3,2,1,0,)(21ˆ=+=n hv n H ψψChapter 051. For the ground state of the one-dimensional harmonic oscillator, compute the standard deviations ∆x and ∆p x and check that the uncertainty principle is obeyed.Answer:1) The ground state wavefunction of the one-dimensional harmonic oscillator is given by2214141)(x e ααπψ--=2) The standard deviations ∆x and ∆p x are defined as222x x x -=∆22∆-∆=p(p∆)(pThe product of ∆x and ∆p is given by2422122 ==∙=∆∆ααp x It shows that the uncertainty principle is obeyed.2. (a) Show that the three commutation relations [x L ˆ,y L ˆ] = z L i ˆ , [y L ˆ,z L ˆ] = x L i ˆ , [z L ˆ,x L ˆ] = y L i ˆare equivalent to the single relation L L Lˆˆˆ i =⨯ (b) Find [2ˆx L,y L ˆ] Answer:1): z y x y x z xz y z y x y x x z z y x y y x z x x z y z z y y z x z z y x y z x y x z y x z y x z y x L i L L L i L L L i L L k L j L i L i k L L j L L i L L k L L L L j L L L L i L L L L i L L j L L i L L k L L j L L k L L k L j L i L k L j L i L L L k L j L i L L ===⇒++=++=-+-+-=-++--=++⨯++=⨯++=],[],[],[)(],[],[],[)()()()()(ˆˆˆ2):)()()(],[],[],[2x z z x xz z x x y x y x x y x L L L L i L L i L i L L L L L L L L L +=+=+=3. Calculate the possible angles between L and the z axis for l = 2.Answer:The possible angles between L and the z axis are equivalent the angles between L and L z . Hence, the angles are given by:Lm Cos L z ==+=θ6)12(2 ︒︒︒︒=7.144,10.114,00.90,91.65,26.35θ4. Complete this equation:m l m l z Y m Y L 333ˆChapter 061. Explain why each of the following integrals must be zero, where the functions are hydrogenlike wave functions: (a) <2p 1|z L ˆ|3p -1>; (b) <3p 0|z L ˆ|3p 0>Answer:Both 3p -1 and 3p 0 are eigenfunctions of L z , with eigenvalues of -1 and 0, respectively. Therefore, the above integrals can be simplified asa) due to orthogonalization properties of eigenfunctions03|213ˆ21111=-=--p p p L p z b) 02. Use parity to find which of the following integrals must be zero: (a) <2s |x |2p x >; (b)<2s |x 2|2p x >; (c) <2p y |x |2p x >. The functions in these integrals are hydrogenlike wave functions.Answer:1) b) and c) must be zero.3. For a hydrogen atom in a p state, the possible outcomes of a measurement of L z are – ħ, 0, and ħ. For each of the following wave functions, give the probabilities of each of these three results: (a)z p 2ψ; (b) y p 2ψ; (c) 12p ψ. Then find <L z > for each of these three wave functions.Answer:a) 022p p z ψψ=, therefore, the probabilities are:0%, 100%, 0% )(2111222-+=p p p x ψψψ, the probabilities are 50%, 0%, 50%.12p ψ,the probabilities are 100%, 0%, 0%b) 0,0,14. A measurement yields 21/2ħ for themagnitude of a particle ’s orbital angular momentum. If L x is now measured, what are the possible outcomes?Answer:1): Since the wavefunction is the eigenfunction of L2, a measurement of the magnitude of the orbital angular momentum should be+L=LL(=⇒21)1,The possible outcomes when measure L x are-1, 0, 1Chapter 071. Which of the following operators areHermitian: d /dx , i (d /dx ), 4d 2/dx 2, i (d 2/d x 2)? Answer :An operator in one-D space is Hermitian if⎰⎰=dx A dx A **)ˆ(ˆψψψψa)⎰⎰⎰⎰-=-=-=∞∞-dxdxd dxdxd dx dx d dx dx d *****)(ψψψψψψψψψψb)⎰⎰⎰⎰=-=-=∞∞-dxdxd i dd i dx dx d i i dx dx d i ****)(ψψψψψψψψψψc)⎰⎰⎰⎰⎰=+-=-=-=∞∞-∞∞-dxdxddddxddxdxddxddxddxddxddxdxdψψψψψψψψψψψψψ2*22****22*44 444 44This operator can be written as a product of 1D kinetic operator and a constant. Hence, it’s Hermitian.d) As the third operator is Hermitian, this operator is not Hermitian.2. If Aˆand Bˆare Hermitian operators, prove that their product AˆBˆis Hermitian if and only if Aˆand Bˆcommute. (b) If Aˆand Bˆare Hermitian, prove that 1/2(AˆBˆ+BˆAˆ) is Hermitian.(c) Is x p xˆˆHermitian? (d) Is 1/2(x p xˆˆ+x p xˆˆ)Hermitian? Answer: 1)If operator A and B commute , we have⎰=-⇒=-⇒=0])ˆˆˆˆ[(0ˆˆˆˆˆˆˆˆˆ*τψψd A B B A A B B A A B B A⎰⎰⎰=⇒=-⇒τψψτψψτψψd A B d B A d A B B A***]ˆˆ[]ˆˆ[0])ˆˆˆˆ[(Operator A and B are Hermitian, we have⎰⎰⎰==⇒τψψτψψτψψd B A d B A d A B ˆˆ)ˆ()ˆ(]ˆˆ[***Therefore, when A and B commute, thefollowing equation fulfills. Namely, AB is also Hermitian.⎰⎰=τψψτψψd B A d B A ˆˆ]ˆˆ[**2)]ˆˆˆˆ[21)]ˆˆˆˆ(21[***⎰⎰⎰+=+τψψτψψτψψd A B d B A d A B B A Operator A and B are Hermitian, we get⎰⎰⎰⎰⎰⎰⎰+=+⇒+=+=+τψψτψψτψψτψψτψψτψψτψψd A B B A d A B B A d B A A B d B A d A B d A B d B A *******])ˆˆˆˆ(21[)ˆˆˆˆ(21])ˆˆˆˆ(21[)ˆˆ()ˆˆ([21]ˆˆˆˆ[21The above equation shows that the operator 1/2[AB+BA] is Hermitian.c) xp x is not Hermitian since both x and px are Hermitian and do not commute. d) YesChapter 081. Apply the variation function cr e -=φto the hydrogen atom; choose the parameter c to minimize the variational integral, and calculate the percent error in the ground-state energy. Solution :1) The requirement of the variation function being a well-behaved function requires that c must be a positive number.2) check the normalization of the variation function.322*)(c d d Sin dr r ed cr πϕθθτφφ==⎰⎰⎰⎰-3) The variation integral equals to)2(214])2[(2)1(21()121(ˆ32223*32*32*3**-=-∂∂+∂∂-=-∇-=-∇-==⎰⎰⎰⎰⎰⎰--c c c dr r e r r r e c d r cc d r cd d H w cr cr τφφπφπτφφπτφφτφφ4) The minimum of the variation integral is21101-=⇒=⇒=-=∂∂w c c c w5) The percent error in the ground state is 0%2. If the normalized variation function x l 2/13)/3(=φ for 0 ≤ x ≤ l is applied to the particle-in-a-one-dimensional-box problem, one finds that the variation integral equals zero, which is less than the true ground-state energy. What is wrong? Solution:The correct trail variation function must be subject to the same boundary condition of the given problem. For the particle in a 1D box problem, the correct wavefunction must equal to zero at x=0 and x=l. However, the trial variation function x l 2/13)/3(=φ does not fulfill these requirement. The variation integral basedon this incorrect variation function does not make any sense.3. Application of the variation function 2cx e-=φ(where c is a variation parameter) toa problem with V = af (x), where a is a positive constant and f (x ) is a certain function of x , gives the variation integral as W = c ħ2/2m+15a /64c 3. Find the minimum value of W for this variation function. Solution:23434123434141min4141413272598.03)25(23)25(0)64152( m a m a w m a c dc c a m c d c w ==⇒±=⇒=+=∂∂4. In 1971 a paper was published that applied the normalized variation functionN exp(-br 2a 02-cr /a 0) to the hydrogen atom and stated that minimization of the variation integral with respect to the parameters b and c yielded an energy 0.7% above the true ground-state energy for infinite nuclear mass. Without doing any calculations, state why this result must be wrong. Solution:From the evaluation of exercise 1, we know that the variation function exp(-cr) gives no error in the ground state of hydrogen atom. This function is a special case of the normalized variation function N exp(-br 2a 02-cr /a 0) when b equals to zero. Therefore, adopting the normalized variation function as a trial variation function should also have no error in the ground state energy for hydrogen atom.5. Prove that, for a system with anondegenerate ground state, 0*ˆE d H>⎰τφφ, if φ is any normalized, well-behaved function that is not equal to the true ground-state wavefunction. (E 0 is the lowest-energy eigenvalueof Hˆ) Solution:As the eigenfunctions of the Hermitian operator H form a complete set, any well-behaved function which is subject to the same boundary condition can be expanded as a linear combination of the eigenfunction of the Hermitian operator, namely,∑∞==0i i i c ψφ, where ψis are eigenfunctions of Hermitian operator H, c i s are constant.The expectation value of φ with respect to the Hermitian operator is20102020102002*00**00*0*0*ˆˆ)(ˆE c E E c E c c E c E cE c c E c cH c c d c H c d H i i i i i ii i ii iii ijj j ji ii j j i i j j j i i i ==+>+======∑∑∑∑∑∑∑⎰∑∑⎰∑∑⎰∞=∞=∞=∞=∞=∞=∞=∞=∞=∞=∞=δψψτψψτφφChapter 09, 101. For the anharmonic oscillator with Hamiltonian43222212ˆdx cx kx dx d m H +++-= , evaluate E (1)for the first excited state, taking the unperturbed system as the harmonic oscillator. Solution:The wavefunction of the first excited state of the harmonic oscillator is241312)4(x xeαπαψ-=Hence, the first order correct to energy of the first excited state is given by6213422134134324131'*1415)4()4()4)(()4(ˆ222απαπαπαπαψψαααd dx ex d dx x d ex xx d x c xedx H x xx ==∙=∙+∙=---⎰⎰⎰⎰2. Consider the one-particle, one-dimensional system with potential-energy V = V 0 forl x l 4341<<, V = 0 forl x 410≤≤ andl x l ≤≤43and V = ∞ elsewhere, where V 0 = 22/ml . Treat the system as a perturbed particle in a box. (a) Find the first-order energy correction for the general stationary state with quantum number n . (b) Find the first-order correction to the wave function of the stationary state with quantum number n . Solution:The wavefunction of a particle in 1D box is given by)(2)0(x ln Sin l nπψ=Take this as unperturbed wavefunction, andthe perturbation H ’ is given by V .a) The first-order energy correction for ψn is])23[]2[(224]2[4[2)()(2)()(2ˆ00004341)0('*)0()1(πππππππππψψn Sin n Sin n V V n S n Sin l l V dx V x l n Sin x l n Sin l dx x ln VSin x l n Sin l dx H E lln n-+=-+====⎰⎰⎰b) The first correction to the wavefunction is given by)0()0()0()0()0()1(2)0()0(222)0('(818mnm mnnm nm n nEEH n E E ml h n Eψψψψ∑∞≠-==-⇒=3. For an anharmonic oscillator with3222212ˆcx kx dx d m H ++-= , take 'ˆHas cx 3. (a) Find E (1) for the state with quantum number v . (b) FindE (2)for the state with quantum number v . You will need the following integral: 3,'2/13)0(3)0('2/)1[(3]8/)3)(2)(1[(||++++++>=<v v v v v v v v x αδαψψ1,'2/38/)2)(1([)2/(3---++v v v v v v αδαSolution:a) As the potential of the unperturbed is a even function, the eigenfunctions of the unperturbed system are either even or odd. The perturbation is an odd function with respect to x. Hence, the first order energy correction is zero.b) The second order energy correction is given by)51212(8]38)2)(1(838)1(338)3)(2)(1([()2(3)21(38)3)(2)(1((''2323333332)0()0(1,2/31,2/33,32)0()0(2)0(3)0()0()0()0()0()0()0()2(++-=--++-++-+++=-+++++++=-=-=∑∑∑∞≠-++∞≠∞≠n n hvc hv n n n hv n hv n hv n n n c E E nn n n n n n c E E cx EEH H En m mn n n n n n n nm mn mnnm mnmn n mnαααααδαδαδαψψψψψψ4. Calculate the angle that the spin vector S makes with the z axis for an electron with spin function α. Solution:For an electron, both S and S z equal to one half. The magnitude of S is74.54]31[23)1(===+=ArcCos S S S θ 5. (a) Show that12ˆP and23ˆP do not commutewith each other. (b) Show that 12ˆPand 23ˆP commute when they are applied to antisymmetric functions. Solution:a) Set the wavefunction to be)1()3()2()2()1()3(321321φφφφφφ≠b) When the function is antisymmetric, we have ψψψψψψψ2312121223231223ˆˆ)(ˆˆˆ)(ˆˆˆP P P P P P P P =-=-==-=-=6. Which of the following functions are (a) symmetric? (b) antisymmetric?(1) )2()1()2()1(ααg f ; (2) )]2()1()2()1()[2()1(αββα-f f ; (3) )3()2()1()3()2()1(βββf f f ; (4) )(21r r a e --;(5) )]1()2()2()1()][2()1()2()1([βαβα--f g g f ; (6) )(21221r r a e r +-. Solution:(2) is antisymmetric(3), (5) and (6) are symmetricChapter 11, 131. How many electrons can be put in each of the following: (a) a shell with principal quantum number n ; (b) a subshell with quantum numbers n and l ; (c) an orbital; (d) a spin-orbital? Solution:a) 2n 2, b) 2*(2l+1), c) 2, d) 12. Give the possible values of the total-angular-momentum quantum number J that result from the addition of angular momentum with quantum numbers (a) 3/2 and 4; (b) 2, 3, and 1/2 Solution:Coupling between two angular momentums with quantum number j 1 and j 2 gives the possible quantum number J of the total angular momentum as:2121j j J j j +<<-a) The possible values are 11/2, 9/2, 7/2, 5/2b) The possible values are:11/2, 9/2, 9/2, 7/2, 7/2, 5/2, 5/2, 3/2, 3/2, 1/23. Find the terms that arise from each of the following electron configurations: (a) 1s22s22p63s23p5g; (b) 1s22s22p3p3d(c) 1s22s22p24dSolution:As fully-filled sub-shells do not contribution the total orbital and spin angular momentum, we can ignore the electrons in these sub-shells while considering the atomic terms. Hence, a) The atomic terms can be:3H, 1H, 3G, 1G, 3F, 1Fb) The atomic terms can be:4G, 2G, 4F, 2F, 4D, 2D, 4P, 2P, 4S, 2S4F 2F, 4D, 2D, 4P, 2P4D, 2D, 4P, 2P, 4S, 2Sc) The atomic terms can be:2G, 2F, 2D, 2P, 2S4F, 2F, 4D, 2D, 4P, 2P。
量子化学考试试题

量子化学考试试题一、选择题(每题 5 分,共 30 分)1、量子化学中,描述微观粒子运动状态的函数被称为()A 波函数B 概率密度C 哈密顿量D 薛定谔方程2、下列哪个量子数决定了原子轨道的形状()A 主量子数B 角量子数C 磁量子数D 自旋量子数3、对于氢原子的 1s 轨道,其电子出现概率最大的位置是()A 原子核处B 离核无穷远处C 离核一定距离处D 无法确定4、量子化学中,计算分子能量常用的方法是()A 半经验方法B 从头算方法C 密度泛函理论D 以上都是5、下列哪种化学键具有明显的量子力学特征()A 离子键B 共价键C 金属键D 氢键6、在量子化学中,分子轨道是由原子轨道线性组合而成,这一原理被称为()A 杂化轨道理论B 价键理论C 分子轨道理论D 晶体场理论二、填空题(每题 5 分,共 30 分)1、量子力学的基本假设包括波函数假设、算符假设、测量假设、全同性原理和__________________ 。
2、氢原子的薛定谔方程在球坐标下的解中,径向波函数 R(r) 与__________________ 有关。
3、多电子原子的电子排布遵循的原则有能量最低原理、泡利不相容原理和__________________ 。
4、分子的偶极矩是衡量分子__________________ 的物理量。
5、密度泛函理论的核心思想是将体系的能量表示为__________________ 的泛函。
6、量子化学计算中,常用的基组有 STO-3G、6-31G 等,其中 6-31G 表示的是__________________ 。
三、简答题(每题 10 分,共 20 分)1、简述量子化学中 HartreeFock 方法的基本思想。
2、解释为什么分子的振动光谱通常具有一系列的吸收峰,而不是单一的吸收峰。
四、计算题(共 20 分)已知氢原子处于某一激发态的波函数为:ψ =1/√8π a₀³(r/a₀) exp(r/2a₀) ,其中 a₀为玻尔半径。
量子化学习题集

(2) x, x2, 3x21; (3) sinx, cosx;
(4) sinx, cosx, tanx;
(5)
sin2x, cos2x, 1;
(6) sinx, cosx, eix (7) sin2x, cos2y, 1
2.2 三维势箱中一粒子的波函数是下列那些算符的本征函数?
ˆ x (2)p ˆx (1) p
x sin bxdx b
1
2
sin ax sin bxdx xe
bx
sin[(a b) x] sin[(a b) x] a 2 b2 2(a b) 2(a b)
x 1 dx 2 ebx b b 2 2x 2 bx x 2 bx 3 x e dx e 2 b b b
2 ˆ+ (3) 2,2|M |2,0; 2ˆ ˆ2 ˆ+ MzM|2,0 (6) 2,0|M
3.9 写出 2p 组态中 3P 谱项的全部波函数
第四章 近似方法
4.1 用尝试变分函数 e b x 求谐振子的基态能级和波函数。 4.2 对一维势箱中粒子应用线性变分函数 c1 x 2 (l x) c2 x(l x) 2 ,计算 n=1 和
2
A
A
2
A2 A ,用一
2
维势箱,验证 x 和 px 的不确定关系。
第三章 角动量、自旋和原子光谱
3.1 对 l=2,计算 L 与 z 轴之间可能的夹角。
2 ˆ 2 ˆx 3.2 证明球谐函数是算符M +M y的本征函数,并求其本征值。 →
ˆ x, M ˆ y]=iM ˆ z [M ˆ y, M ˆ z]=iM ˆ x [M ˆ z, M ˆ x]=iM ˆy 3.3 证明角动量的三个对易规则 [M ˆ = iM ˆ ˆ M 可合并写成 M ˆ x, M ˆ y, M ˆ z 为厄米算符。 3.4 证明:M ˆ ,G ˆ ]0),设为体系的某一状态,则有 3.5 如果两个力学量算符不对易([F
量子化学期末考试试卷及参考答案
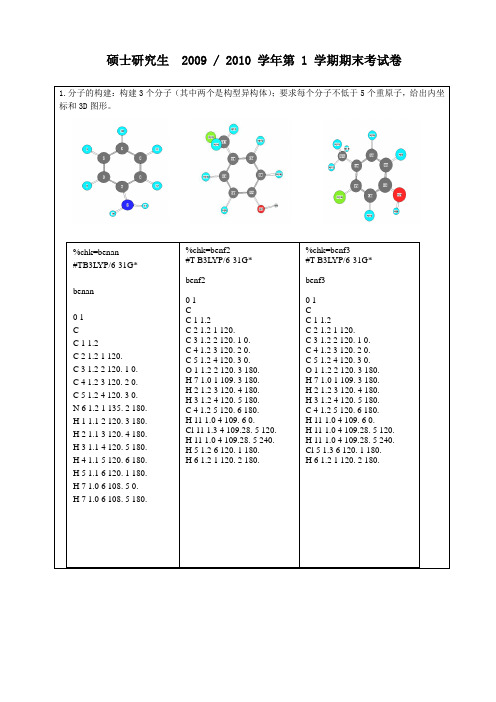
优化结构后的键长: C1-C2=1.399 C2-C3=1.388 C3-C4=1.407 C5-C6=1.398 C1-C6=1.395 C2-H9=1.085 C1-O7=1.366 O7-H8=0.970 C3-H10=1.088 C4-C11=1.511 C11-H12=1.091 C11-H13=1.097 C11-H14=1.097 C5-Cl15=1.786 C6-H16=1.086
3-氯代对甲基苯酚优化后的能量是:HF=-806.3769679 优化后的构型如下: HOMO
LUMO
各原子所带的电荷: Total atomic charges:
1 C 0.357296 2 C -0.150569 3 C -0.180892 4 C 0.171258 5 C -0.134553 6 C -0.194460 7 O -0.640742 8 H 0.410639 9 H 0.144774 10 H 0.133768 11 C -0.533938 12 H 0.180060 13 H 0.164373 14 H 0.164373 15 Cl -0.032464 16 H 0.141076 Sum of Mulliken charges= 0.00000
%chk=benf3 #T B3LYP/6-31G*
benf3
01 C C 1 1.2 C 2 1.2 1 120. C 3 1.2 2 120. 1 0. C 4 1.2 3 120. 2 0. C 5 1.2 4 120. 3 0. O 1 1.2 2 120. 3 180. H 7 1.0 1 109. 3 180. H 2 1.2 3 120. 4 180. H 3 1.2 4 120. 5 180. C 4 1.2 5 120. 6 180. H 11 1.0 4 109. 6 0. H 11 1.0 4 109.28. 5 120. H 11 1.0 4 109.28. 5 240. Cl 5 1.3 6 120. 1 180. H 6 1.2 1 120. 2 180.
量子化学期末考试及答案

量子化学期末考试及答案量子化学期末考试一、选择题(每题2分,共20分)1. 量子化学的基本概念是:A. 原子的能量状态B. 原子的电子结构C. 原子的结构D. 原子的电荷2. 在量子化学中,电子的状态可以用:A. 原子轨道表示B. 原子能量表示C. 原子电荷表示D. 原子结构表示3. 在量子化学中,电子的状态可以用:A. 原子轨道表示B. 原子能量表示C. 原子电荷表示D. 原子结构表示4. 在量子化学中,电子的状态可以用:A. 原子轨道表示B. 原子能量表示C. 原子电荷表示D. 原子结构表示5. 在量子化学中,电子的状态可以用:A. 原子轨道表示B. 原子能量表示C. 原子电荷表示D. 原子结构表示二、填空题(每题2分,共20分)1. 量子化学的基本原理是____________。
答:量子力学。
2. 量子化学中,电子的状态可以用____________表示。
答:原子轨道。
3. 量子化学中,电子的能量可以用____________表示。
答:原子能量。
4. 量子化学中,电子的电荷可以用____________表示。
答:原子电荷。
5. 量子化学中,电子的结构可以用____________表示。
答:原子结构。
三、论述题(每题10分,共30分)1. 请简要介绍量子化学的基本原理。
答:量子化学是一门研究原子和分子的科学,它基于量子力学的原理,用来描述原子和分子的结构和性质。
量子力学的基本原理是,电子的运动是由量子态决定的,而不是由经典物理学的运动方程决定的。
量子力学的基本原理是,电子的运动是由量子态决定的,而不是由经典物理学的运动方程决定的。
量子力学还提出了一种新的概念,即电子的状态可以用原子轨道、原子能量、原子电荷和原子结构等表示。
量子力学的基本原理是,电子的运动是由量子态决定的,而不是由经典物理学的运动方程决定的。
2. 请简要介绍量子化学的应用。
答:量子化学的应用非常广泛,它可以用来研究原子和分子的结构和性质,以及它们之间的相互作用。
应化所量子化学考试题库及其答案详解

Principles Of Quantum Chemistry——Kwong.S.T名词解释1.测不准关系:()()41M L 22≥∆⋅∆{∫ψ*i[L ,M ]ψdx}22.酉矩阵:S +=S -13.厄米算符:算符L 满足∫u 1*(x)L u 2(x)dx=∫u 2(x)L *u 1*(x)dx,其中u 1(x)和u 2(x)是任意两个平方可积函数,积分遍于自变量全部区域。
则称L 是厄米算符。
4.等价表示:矩阵群M 和M’所包含的每一个对应矩阵之间只差一个同样的相似变换,就说M’和M 是等价表示。
5.可约表示:如果一个矩阵表示可以表示成子矩阵的直和,那么这个矩阵表示是可约表示。
6.不可约表示特征标:不可约表示矩阵群的对角元素之和称为这个不可约表示的特征标。
7.投影算符:P R R hl k j Rj j k*∑Γ=λλ)()()(,其中R 为群元素,*Γk j R λ)()(是第j 个不可约表示操作R 的第λ行第k 列的矩阵元,l j 是第j 个不可约表示的维数,h 为群的阶。
8.轨道近似:认为各个电子的运动是彼此独立的,每个电子都在核与其它电子所形成的稳定的平均场中运动,从而每个电子的状态可以用一个单电子波函数来描写。
9.定域分子轨道:认为成键电子只是集中在相邻两原子间的键轴区域内。
10.正则分子轨道:由HFR 方程解出的分子轨道。
11.Slater 行列式:描述多电子体系满足保里原理的波函数。
12.ab initio :严格的按HFR 方程进行计算称为从头计算。
13.NDO :假设原子轨道在空间任何地方都不重叠。
DO :对所有不同原子轨道的乘积都采用忽略微分重叠近似。
15.NDDO :对属于同一原子各对轨道重叠的排斥积分全部予以保留。
16.INDO :只保留单中心积分中同一原子对各轨道的微分重叠。
17.EHMO :整个分子不在一个平面上,则σ,π分离就不可能,于是就必须对分子中所有原子的所有价电子进行计算,若这种计算不是基于自洽场分子轨道理论,就称之为推广的休克尔分子轨道法。
量子化学习题及答案

量子化学习题及答案1.1998及2013年度诺贝尔化学奖分别授予了量子化学以及分子模拟领域的杰出贡献者,谈谈你的了解及认识。
答:1998年诺贝尔化学奖得主:瓦尔特·科恩和约翰·波普尔。
1964-1965年瓦尔特·科恩提出:一个量子力学体系的能量仅由其电子密度所决定,这个量比薛定谔方程中复杂的波函数更容易处理得多。
他同时还提供一种方法来建立方程,从其解可以得到体系的电子密度和能量,这种方法称为密度泛函理论,已经在化学中得到广泛应用,因为方法简单,可以应用于较大的分子。
沃尔特·库恩的密度泛函理论对化学作出了巨大的贡献。
约翰·波普尔发展了化学中的计算方法,这些方法是基于对薛定谔方程中的波函数作不同的描述。
他创建了一个理论模型化学,其中用一系列越来越精确的近似值,系统地促进量子化学方程的正确解析,从而可以控制计算的精度,这些技术是通过高斯计算机程序向研究人员提供的。
今天这个程序在所有化学领域中都用来作量子化学的计算。
2013年诺贝尔化学奖得主:马丁·卡普拉斯、迈克尔·莱维特、阿里耶·瓦谢勒。
他们为复杂化学系统创立了多尺度模型。
为研发了解和预测化学过程的强有力的计算机程序奠定了基础。
对于今天的化学家来说,计算机就像试管一样重要。
模拟过程是如此的真实以至于传统实验的结果也能被计算机预测出来。
多尺度复杂化学系统模型的出现无疑翻开了化学史的“新篇章”。
化学反应发生的速度堪比光速。
刹那间,电子就从一个原子核跳到另一个原子核,以前,对化学反应的每个步骤进行追踪几乎是不可能完成的任务。
而在由这三位科学家研发出的多尺度模型的辅助下,化学家们让计算机做“做帮手”来揭示化学过程。
20世纪70年代,这三位科学家设计出这种多尺度模型,让传统的化学实验走上了信息化的快车道。
2.谈谈你对量子化学中两种流派(VBT,MOT)的认识。
答:1926年,奥地利物理学家薛定谔(Schrodinger)建立了描述电子运动规律的波动方程。
量子化学试题及答案

6-31G*=6-31G(d)6:代表每个内层轨道由六个高斯型基函数拟合而成;价层轨道劈裂成两个Salter型基函数,内层轨道不发生劈裂,其中一个Salter型基函数由一个Gauss型基函数拟合而成,另一个Salter型基函数由一个Gauss型基函数拟合而成;d:表示要对出氢以外的原子都要加d轨道Salter型基函数:2*4+(1+2+2*3+6)*2=38Gauss型基函数:4*4+(6+4+4*3+1*6)*2=722、解:第一种方法:CH,1, 1.08290068H,1, 1.08290068,2, 109.47122063H,1, 1.08290068,2, 109.47122063,3,120.0,0H,1, 1.08290068,2, 109.47122063,3,-120.0,0第二种方法:CH 1 B1H 1 B2 2 A1H 1 B3 2 A2 3 D1 0H 1 B4 2 A3 3 D2 0B1 1.08290068B2 1.08290068B3 1.08290068B4 1.08290068A1 109.47122063A2 109.47122063A3 109.47122063D1 120.00000000D2 -120.000000003、解:在分子势能面上有五类极值点,分别如下:整体极小点、局部极小点、整体极大点、局部极大点及鞍点。
整体极小点:整个势能面上的最低点,代表了能量最低也就是最稳定的结构;局部极小点:势能面某个区域内的最低点,代表了局部区域内能量最低的点;整体极大点:整个势能面上的最高点,代表了能量最高的点;局部极大点:势能面某个区域内的最高点,代表了局部区域内的能量最高的点;鞍点:在一个方向上是极小点,其他方向上都是极大点,代表了体系的过渡态。
判断某一极值点是否为过度态:首先,是否有且只有一个虚频(数值为负值,足够大,一般上百);其次,看虚频的震动模式是不是朝着反应物和产物的方向震动;再次,进行IRC计算,看看是不是总想了反应物和产物。
- 1、下载文档前请自行甄别文档内容的完整性,平台不提供额外的编辑、内容补充、找答案等附加服务。
- 2、"仅部分预览"的文档,不可在线预览部分如存在完整性等问题,可反馈申请退款(可完整预览的文档不适用该条件!)。
- 3、如文档侵犯您的权益,请联系客服反馈,我们会尽快为您处理(人工客服工作时间:9:00-18:30)。
群论与量子化学期中考试
姓名:专业:学号:
一、判断下列集合是否够成群。
(4分)
(1){除零以外的全部有理数}结合规则是乘法y (2){1,0,-1 },结合规则是加法n
(3){1,0,-1 },结合规则是乘法n
(4){1,-1,i,-i},结合规则是乘法y
二、判断分子所属点群。
(27分,每个0.5分)
三、假定CuCl 42-原来属于T d 点群,四个Cl 原子的标号如图所示,当出现以下情况是,它所属的点群如何变化。
(10分) (1)Cu —Cl (1)键长缩短; C 3v
(2)键长Cu —Cl (1)和Cu —Cl (2)缩短相同长度;C 2v (3)键长Cu —Cl (1)和Cu —Cl (2)缩短不同长度;Cs (4) Cl (1)—Cl (2)和Cl (3)—Cl (4)间距离缩短不同的长度;C 2v (5)Cl (1)—Cl (2)和Cl (3)—Cl (4)间距离缩短相同的长度。
D 2d
注:若Cu —Cl 键缩短,假定Cu 原子不动,只有相应的Cl 原子移动, 若Cl —Cl 间的距离缩短,视为两个原子分别向两原子连线的中
心移动相同的距离。
四、证明:同一类操作矩阵的特征标相等。
(6分) 证明:Tr(R)=Tr(S -1R’S )=Tr(S S -1 R’)=Tr(R’).命题得证。
五、用D 3d 群的特征标表验证小正交定理。
(5分)
()*()
()*()()*()()()R
i i i i
i j j ij
R R g g g g g νμμννμμνννν
χχδχχδχχδ===∑∑∑
六、试求具有如下的阶和类数的群的不可约表示的维数。
(6分) (1)6阶,3类 1 1 2 (2)12阶,4类 1 1 1 3 (3)24阶,5类 1 1 2 3 3
七、将下列可约表示的特征标写成不可约表示特征标的直和。
(10分) (1) C 2v 群 E C 2 σv σv ’ Γ 5 -3 3 -1
1212112
1
a(A )=(51313111)1
41
a(A )=(51313111)0
41
a(B )=(51313111)3
41
a(B )=(51313111)1
4
A 3
B B ⨯-⨯+⨯-⨯=⨯-⨯-⨯+⨯=⨯+⨯+⨯+⨯=⨯+⨯-⨯-⨯=Γ=⊕⊕ (2)
C 3v 群 E 2C 3 3σv Γ 292 -119 8
12121
()(292121191381)13
61
()(292121191381)5
6
1
()(292221191)137
6
135137a A a A a E A A E
=⨯-⨯⨯+⨯⨯==⨯-⨯⨯-⨯⨯==⨯+⨯⨯=Γ=⊕⊕ 八、求C 3v 群的13A 1⊕46A 2⊕287E 表示的χ(2
3ˆC )。
(10分)
χ(23C )=13×1+46×1-287×1=-228
九、在分子PF 5中,对于存在的不可约表示''2A ,利用投影算符P(''2A ) 作用在p z1,求投影后得到的基。
(10分)
''21''21
112221121ˆˆ()()()1
(2323)121
()2
z A p R z R
z z z z z z z z P A R O p h p p p p p p p p ϕχ==+++++=+∑ 十、反式N 2H 2。
(12分) (1)判断分子所属点群。
(2)写出分子的振动频率的数目。
(3)以每个原子的三个直角坐标作为可约表示的基,写出分子所属
点群的可约表示(以分子平面为xy 平面)。
(4)根据特征标表将分子的可约表示化为不可约表示。
(5)找出分子的平动、转动和振动的不可约表示,并判断振动的红外和拉曼活性。
(6)找出属于分子平面内的振动所属的不可约表示。
(1)C2h(2)3×4-6=6 (3)Γ=12E⊕4σh
(4)Γ=4A g⊕2B g⊕2A u⊕4B u
(5)Γtrans=A u⊕2B uΓrot=A g⊕2B gΓvib=3A g⊕A u⊕2B u
A g有拉曼活性,A u和
B u有红外活性
(6)属于A g和B u的不可约表示的振动在分子平面内
附:考试中可能会使用到的特征标表。
